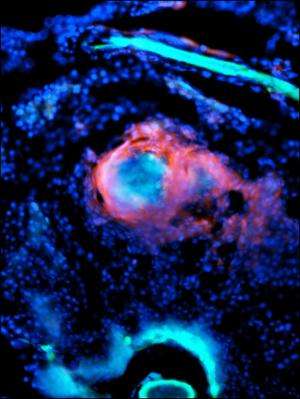
When zebrafish are infected with Mycobacterium marinum (blue), immune cells (red) quickly surround the bacteria to form tightly organized nuggets called granulomas. Within days, blood vessels (green) begin to sprout in and around these little balls of cells. Credit: Stefan Oehlers
The body responds to tuberculosis infection by locking the bacterial offenders into tiny clusters of immune cells called granulomas, which are a hallmark of the disease. This containment strategy succeeds at first, but eventually the bacteria manage to break out of these intercellular jails and spread throughout the body.
Now, researchers have shown that this escape is aided by new blood vessels that tunnel into the granulomas, providing fresh oxygen and a way out for the bacteria held inside. They tested FDA-approved drugs that block this process—known as angiogenesis—and found they could effectively reduce the numbers of bacteria, limit their spread outside of granulomas, and increase the survival of infected laboratory animals.
The findings, which appear online Nov. 24 in Nature, suggest a new therapeutic approach that might target the body's response to tuberculosis (TB) rather than going after the bacteria directly.
"Angiogenesis is a host response that can be triggered by granulomas, and the bacteria take advantage of it to grow and to maintain a sufficient supply of oxygen, and ultimately to spread," said David M. Tobin, Ph.D., an assistant professor of molecular genetics and microbiology at Duke University School of Medicine.
"We think that anti-angiogenic agents, commonly used for cancer, could be an entirely new avenue for treating TB," Tobin said. "This sort of host-directed therapy would be particularly effective in cases of multi-drug-resistant disease where current antibiotic regimens fail."
Tuberculosis is a leading cause of death from infectious diseases worldwide, second only to HIV/AIDS. A recent report from the World Health Organization found that 9 million people developed TB and 1.5 million died from the disease in 2013 alone. Though there are several drug regimens approved to treat the illness, these treatments can be time-consuming, toxic, and ineffective against the most drug-resistant strains.
When zebrafish are infected with bright blue Mycobacterium marinum, bright red immune cells quickly surround the bacteria to form tightly organized nuggets called granulomas (vessels green, bacteria blue, immune cells red). Within days, bright green blood vessels are seen sprouting in and around these little balls of cells. The new vessels are functioning blood vessels carrying red blood cells in the same way as normal vessels. Credit: Stefan Oehlers
Researchers have pursued alternative therapies for TB using a variety of animal models, including the zebrafish found in many pet stores and household aquariums. The tiny fish can be infected by its natural pathogen Mycobacterium marinum, a close cousin to Mycobacterium tuberculosis, the bacterium that causes human TB.
Obviously, the fish don't have lungs, but they do develop granulomas that are structurally similar to those found in human TB patients.
Tobin, who helped to develop the zebrafish as a model of TB, decided to use the system to study the blood vessels associated with granulomas. He and his postdoctoral fellow, Stefan H. Oehlers, Ph.D., wanted to determine whether these new blood vessels were benefiting the host or the bacteria.
Because zebrafish are transparent until their stripes emerge in adulthood, Oehlers could label the bacteria, immune cells, and vasculature with differently colored fluorescent tags and then follow a TB infection over time. When he infected zebrafish with bright blue Mycobacterium marinum, he saw bright red immune cells quickly surround the bacteria to form tightly organized granulomas. Within days, bright green blood vessels could be seen sprouting in and around these little balls of cells. They found that the new vessels were functioning blood vessels carrying red blood cells in the same way as normal vessels.
The researchers wondered what would happen if the new blood vessels were kept from forming. If they were important to the host, then blocking this process would only make the infection worse. But if their purpose was to help the bacteria grow and spread, then blocking the process could help contain the infection. To differentiate the two possibilities, the researchers dropped a dose of the anti-angiogenesis drugs semaxinib and pazopanib—already used to treat cancer—into the zebrafish's water.
They found that these drugs suppressed the growth of new blood vessels, reduced the numbers of bacteria, and limited the spread of TB outside the granulomas. Importantly, treatment with the popular anti-cancer drug pazopanib tripled the survival rate of zebrafish with TB. The anti-angiogenesis drugs also increased the effectiveness of rifampicin, a first-line TB therapy.
"We think that cutting off vessel growth to the tuberculosis bacteria effectively strangles the pathogen and makes it an easier target for the immune system," Stefan Oehlers said. "Now we would like to show that targeting host blood vessels is effective in other preclinical tuberculosis models."
More information: "Interception of host angiogenic signaling limits mycobacterial growth," Stefan H. Oehlers, Mark R. Cronan, Ninecia R. Scott, Monica I. Thomas, Kazuhide S. Okuda, Eric M. Walton, Rebecca W. Beerman, Philip S. Crosier, and David M. Tobin. Nature, Advance Online, Nov. 24, 2014. DOI: 10.1038/nature13967
Journal information: Nature
Provided by Duke University