Overactive bladder a common problem, FDA says
(HealthDay)—More than 33 million Americans suffer from overactive bladder, including 40 percent of women and 30 percent of men, the U.S. Food and Drug Administration says.
There are numerous approved treatments for the condition, but many people don't seek help because they're embarrassed or don't know about therapy options, according to an agency news release.
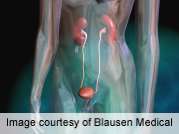
In people with overactive bladder, the bladder muscle squeezes too often or squeezes without warning. This can cause symptoms such as: the need to urinate too often (eight or more times a day, or two or more times a night); the need to urinate immediately; or accidental leakage of urine.
Treatments for overactive bladder include oral medications, skin patches or gel, and bladder injections.
"There are many treatment options for patients with overactive bladder. Not every drug is right for every patient," Dr. Olivia Easley, a senior medical officer with the FDA Division of Bone, Reproductive and Urologic Products, said in the FDA news release.
"Patients need to take the first step of seeking help from a health care professional to determine whether the symptoms they are experiencing are due to overactive bladder or another condition, and to decide which treatment is the best," Easley added.
Anticholinergics are a class of medications widely used to treat overactive bladder. These drugs are believed to work by inhibiting involuntary bladder contractions, she explained.
A recently approved drug, called Myrbetriq (mirabegron), improves the bladder's ability to store urine by relaxing the bladder muscle during filling.
For women aged 18 and older with overactive bladder, an over-the-counter patch called Oxytrol for Women can be applied to the skin every four days. A skin patch for men is available by prescription only.
Botox injections are another option. The Botox is injected directly into the bladder muscle, causing it to relax and increase its ability to store urine. Injections can be given a minimum of three months apart. However, Botox may cause serious and potentially life-threatening side effects such as breathing and swallowing problems, according to the FDA.
More information: The U.S. National Library of Medicine has more about overactive bladder.
Copyright © 2015 HealthDay. All rights reserved.