May 12, 2015 report
Study reveals why prolonged light exposure leads to weight gain

(Medical Xpress)—A study conducted by a combined team of researchers from Leiden University Medical Center and the Academic Medical Center, both in The Netherlands, has found the underlying cause of weight gain in mice exposed to a long periods of light. In their paper published in Proceedings of the National Academy of Sciences, the team describes how they tested the food burning efficiency of mice exposed to different amounts of light over an extended period of time and what it revealed about weight gain.
Scientists have known for some time that overexposure to light, or light exposure during odd hours, can cause people to gain weight, contributing to the many maladies that come along with it—but why that happens has not been understood, until now. Many people, particularly those who work at night, assume that their weight gain is due to the added stress they are feeling due to long work hours and an irregular schedule. In this new effort, it appears such people may be right, but not in a way that has been understood.
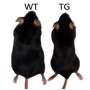
To find out why people (or in this case mice) gain weight if they are exposed to light for many hours each day, the team placed lab mice in enclosures where the day/night duration was modified—different groups lived with artificial daylight lasting for 12, 16 and even 24 hours for over a month. Suspecting that brown adipose tissue (BAT) was playing a part, the team monitored its efficiency in all of the mice. BAT is tissue that plays a significant role in converting energy from food into heat. If energy from food is not converted to heat, it is instead stored as fat.
In looking at the efficiency levels of the BAT, the team found that the mice that were exposed to longer amounts of light, not only gained weight (despite having the same amount of food and exercise as mice not exposed to long light times) but their BAT efficiency declined. Thus, long duration light periods, caused the body to store more food energy, rather than release it as heat. The team suggests this likely applies to people as well and goes back to our early history where summer time (with more daylight hours) meant storing up fat to keep us warm in cold winters.
More information: Prolonged daily light exposure increases body fat mass through attenuation of brown adipose tissue activity, Sander Kooijman, DOI: 10.1073/pnas.1504239112
Abstract
Disruption of circadian rhythmicity is associated with obesity and related disorders, including type 2 diabetes and cardiovascular disease. Specifically, prolonged artificial light exposure associates with obesity in humans, although the underlying mechanism is unclear. Here, we report that increasing the daily hours of light exposure increases body adiposity through attenuation of brown adipose tissue (BAT) activity, a major contributor of energy expenditure. Mice exposed to a prolonged day length of 16- and 24-h light, compared with regular 12-h light, showed increased adiposity without affecting food intake or locomotor activity. Mechanistically, we demonstrated that prolonged day length decreases sympathetic input into BAT and reduces β3-adrenergic intracellular signaling. Concomitantly, prolonging day length decreased the uptake of fatty acids from triglyceride-rich lipoproteins, as well as of glucose from plasma selectively by BAT. We conclude that impaired BAT activity is an important mediator in the association between disturbed circadian rhythm and adiposity, and anticipate that activation of BAT may overcome the adverse metabolic consequences of disturbed circadian rhythmicity.
© 2015 MedicalXpress.com