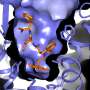
The most powerful analgesic and addictive properties of opiate alkaloids are mediated by the m-opioid receptor (mOR). As the receptor primarily responsible for the effects of opium, the mOR is one of the oldest drug targets within the pharmacopeia.
Opioid receptors are highly versatile signaling molecules. Activation of the mOR results in signalling through the heterotrimeric G protein Gi, resulting in analgesia and sedation as well as euphoria and physical dependence3. The mOR can also signal through arrestin, and this pathway has been attributed to adverse effects of opioid analgesics including tolerance, respiratory suppression, and constipation.
An international team of scientists amongst whom Jan Steyaert and Toon Laeremans (VIB/Vrije Universiteit Brussel) have unraveled the structural basis for mORactivation. In two Nature papers, they report on the X-ray and NMR structures of the agonist-bound mOR receptor, stabilized in its fully activated state by use of a G protein mimetic camelid antibody fragment.
"This knowledge may pave the way to new morphine-like pain killers with fewer side effects," says researcher Jan Steyaert.
More information: "Structural insights into m-opioid receptor activation." Nature 2015 - DOI: 10.1038/nature14886
"Propagation of conformational changes during m-opioid receptor activation." Nature 2015 - DOI: 10.1038/nature14680
Journal information: Nature
Provided by VIB (the Flanders Institute for Biotechnology)