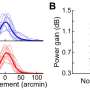
Age-related macular degeneration (AMD) is the leading cause of visual impairment in people over 50 in developed countries. Although this condition affects up to 20% of people between the ages of 65 and 75, what drives abnormal blood vessels to invade the retina is unknown. Researchers from Montreal and Boston now provide a new mechanism for that blinding retinal disease in a study just published in the prestigious scientific journal Nature Medicine.
"In a murine model of AMD, we found that the inability of photoreceptors - nerve cells that capture light and generate vision - to produce energy may drive abnormal blood vessels to invade the retina" said Jean-Sébastien Joyal, an intensive care pediatrician at mother-child research hospital CHU Sainte-Justine and professor in the Department of Pediatrics at the University of Montreal. "We also discovered that photoreceptors do not rely exclusively on glucose to produce energy as previously thought, but also use lipids as a fuel substrate (like the heart, for example)" he said.These findings debunk the scientific beliefs surrounding the cause of abnormal proliferation of blood vessels leading to blindness, and thus open new therapeutic avenues for retinal diseases such as AMD.
Compensating for energy deficiency
The retina is covered with photoreceptors, which thrive on oxygen and nutrients carried by the blood. The abnormal growth of blood vessels causes blindness in some retinal diseases. "In general, the scientific community agrees that proliferation of blood vessels occurs when the body attempts to compensate for oxygen deficiency. We now show that abnormal vessels may also grow to compensate for a lack of fuel or energy production, in a model of AMD," said Dr. Joyal.
Fat as energy substrate—a possible evolutionary advantage
The type of energy used also surprised the scientists in their study. "To our surprise, we found that photoreceptors also feed on fatty acids. It was previously believed that these specialized nerve cells, which are energy-intensive, rely primarily on glucose," said Dr. Joyal. The ability of photoreceptors to use different fuel sources to produce energy may provide an evolutionary advantage during feast and famines.
Plunged into darkness to boost energy consumption
The researchers suspected that dysregulated energy metabolism of photoreceptors might contribute to AMD. They used a murine model unable to use lipids efficiently and observed the presence of abnormal retinal blood vessels resembling AMD. These models developed a lot more retinal lesions when raised in darkness, which is known to increase the energy consumption of the retina. This evidence suggested a link between energy demands and vascular supply of the retina.
Lipid sensors control glucose uptake and energy production
When deprived of fatty acids, can photoreceptors use glucose instead? "Probably in normal conditions, but paradoxically not in our lipid-uptake deficient model. These have elevated circulating blood levels of fatty acids. To explain this puzzling observation, we found the presence of lipid sensors on the surface of photoreceptors. We think these receptors help match available fuel substrate in circulation with the energy production of photoreceptors. When lipid sensors detect excess fatty acids in the blood, it considers the available lipid fuel to be sufficient and suppresses glucose absorption."
Dr. Joyal summarizes the situation as follows: "In lipid-uptake deficient models, photoreceptors end up being starved for both fatty acids and glucose. Starved photoreceptors secrete signals that attract new blood vessels in order to increase nutrient supply. By proliferating behind the retina however, these vessels cause a decline in eyesight, leading to blindness." So at the end of the day, what's the take-home message? "Well, there are three based on our in vivo findings", recaps Dr. Joyal. "AMD may be caused in part by an energy deficiency. Fat is also a source of energy for photoreceptors. And lipid sensors may control glucose entry in the retina."
Further work must be carried out to develop new therapy that will build on these discoveries with the aim of preventing, slowing down or reversing vision loss caused by retinal diseases such as AMD.
More information: The article "Retinal lipid and glucose metabolism dictates angiogenesis through lipid sensor Ffar1" was published in the journal Nature Medicine on March 14, 2016. DOI: 10.1038/nm.4059
Journal information: Nature Medicine
Provided by University of Montreal