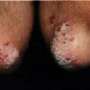
Ixekizumab efficacious for psoriasis over 60 weeks
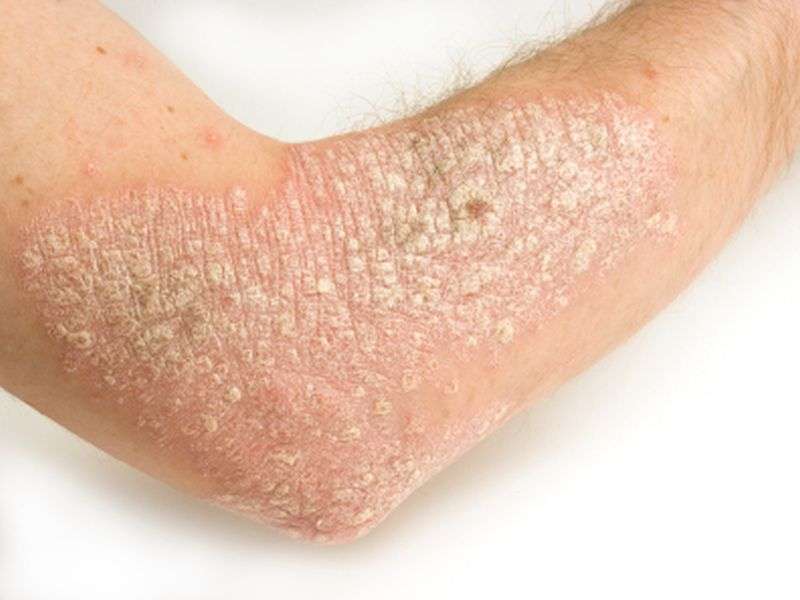
(HealthDay)—The benefits of ixekizumab in the treatment of psoriasis extend to 60 weeks, according to a study published online June 8 in the New England Journal of Medicine.
The findings are based on 3,866 patients who took part in three trials. In one trial, ixekizumab was tested against a placebo; in the other two, it was tested against a placebo and etanercept for the first 12 weeks, and then just placebo from then on. After the initial 12-week period, patients took the drug either once a month or every 12 weeks.
The researchers found that almost three-quarters of patients in the once-a-month group had minimal psoriasis, based on doctors' ratings. That compared with 7 percent of placebo patients. About four out of five patients who took the drug monthly had at least a 75 percent improvement in their skin symptoms. In the initial 12-week study, ixekizumab also outperformed etanercept by a significant margin. The researchers reported adverse events, including neutropenia, candidal infections, and inflammatory bowel disease, with ixekizumab use.
"As with any treatment, the benefits need to be weighed against the risks of adverse events," the authors write. "The efficacy and safety of ixekizumab beyond 60 weeks of treatment are not yet known."
The study was funded by Eli Lilly, the manufacturer of ixekizumab.
More information:
Abstract
Full Text
Copyright © 2016 HealthDay. All rights reserved.