Composite biomaterial scaffolds enable patterning of tissue architecture and cell identity
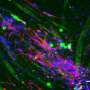
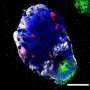
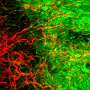
Three-dimensional culture of stem cells in biomaterials has recently enabled the formation of complex cellular structures and miniature organoid tissues, including tissues resembling brain, spinal cord, retina, liver, and kidney. In order to improve this technology further, research published in the Journal of Tissue Engineering describes new designs for unique biomaterial scaffolds that incorporate patterned architectures and regional compartments of signaling factors that can more intricately guide tissue development. These designs enable more comprehensive control over cell fate and tissue architecture, and also establish a platform for studying the effects of concentration gradients of a variety of signaling factors on tissue development.
The ability to form specific molecular concentration gradients within tissue cultures provides several unique advantages and capabilities. Dr. Richard J. McMurtrey, author of the work, said, "The compartments of signaling factors that are designed into the synthetic tissue constructs can form concentration gradients as a result of natural diffusion behaviors, and these gradients can control numerous processes like stem cell differentiation, regional identity, axis patterning, and tissue architecture." As examples, Dr. McMurtrey describes how regional gradients of sonic hedgehog protein (SHH), wnt protein (WNT), bone morphogenic protein (BMP), fibroblast growth factor (FGF), retinoic acid (RA), and reelin protein (RELN) can influence the formation of the nervous system in both innate neural tissue and in three-dimensional (3D) organoids. By separating factors into localized molecular gradients, researchers can mimic developmental cues and can thereby pattern ventral/dorsal and rostral/caudal aspects of the organoid tissue.
Restoring function in damaged neural tissue is likely to require stem cells that can go through similar developmental processes as they did during early development in the womb. "The ability to replicate natural developmental processes in 3D culture of patient-derived stem cells is essential for creating targeted regeneration of specific areas of the brain and spinal cord," said Dr. McMurtrey. "Although there is much we have discovered about how neurodevelopment occurs, there is still much more to learn about all the detailed and complex neurodevelopmental processes that form the vast array of regions, structures, and functions in the brain and spinal cord, and these new tissue designs will help expand our capabilities to study and control these complex processes."
Importantly, the tissue construct designs presented in the paper also enable an array of important investigations in tissue development, disease mechanisms, drug toxicologies, as well as regenerative medicine applications. The combination of biomaterials with stem cells can provide many advantages over stem cell applications alone, including improved cell survival, improved guidance of differentiation processes, improved cellular integration into host tissue, improved control of tissue patterning, and improved migration and sprouting of neural connections. Nevertheless, much research still remains to be done on the optimal combinations of biomaterials, signaling factors, and scaffolding architectures needed to optimally prepare cells for transplantation and integration into specific tissues of the body, and it is hoped that this technology will someday provide capabilities to guide reconstruction of neural architecture in the human nervous system. Dr. McMurtrey stated, "It is a privilege to work on research that is both fascinating and imperative for treating neurological injuries that we simply cannot heal today."
More information: R. J. McMurtrey, Multi-compartmental biomaterial scaffolds for patterning neural tissue organoids in models of neurodevelopment and tissue regeneration, Journal of Tissue Engineering (2016). DOI: 10.1177/2041731416671926