Mouse fatty liver. Credit: C. Maíllo, IRB Barcelona
A team co-headed by scientists at the Institute for Research in Biomedicine and the IDIBAPS Biomedical Research Institute has revealed the capacity of the CPEB4 protein to prevent fatty liver disease.
This condition generally leads to chronic inflammation (non-alcoholic steatohepatitis), which can trigger fibrosis, cirrhosis and ultimately liver cancer. This study on the basic biology of the liver paves the way to examine therapeutic strategies to fight and prevent fatty liver disease. The results have appeared in Nature Cell Biology this week.
CPEB4 and fatty liver
Non-alcoholic fatty liver is characterised by the accumulation of fat deposits in hepatocytes. The development of this condition is determined by many factors that have not been well described to date. However, obesity and lifestyle, as well as aging, are associated with an increase in the incidence of this disease. Also, a number of large-scale genomics studies have linked variants of the CPEB4 gene with the impairment of fat metabolism.
The scientists at IRB Barcelona depleted CPEB4 expression in mouse livers in order to study the function of this protein. They observed that the mice developed fatty liver as they aged. Furthermore, young CPEB4-depleted mice fed a high-fat diet also developed this condition in a more pronounced manner.
Carlos Maíllo, first author of the article and PhD student at IRB Barcelona has described the molecular function of CPEB4. He reveals that this protein is essential to drive the liver stress response.
Specifically, under stress, such as that caused by uncontrolled ingestion of fats, for example, the endoplasmic reticulum—a cell organelle associated with protein synthesis and folding and lipid metabolism—stops its activity in order to re-establish cell equilibrium. This "clean-up" mechanism is orchestrated by CPEB4 and varies in function of the time of day—being more active in humans during the day (when the liver has most work) and dropping off at night.
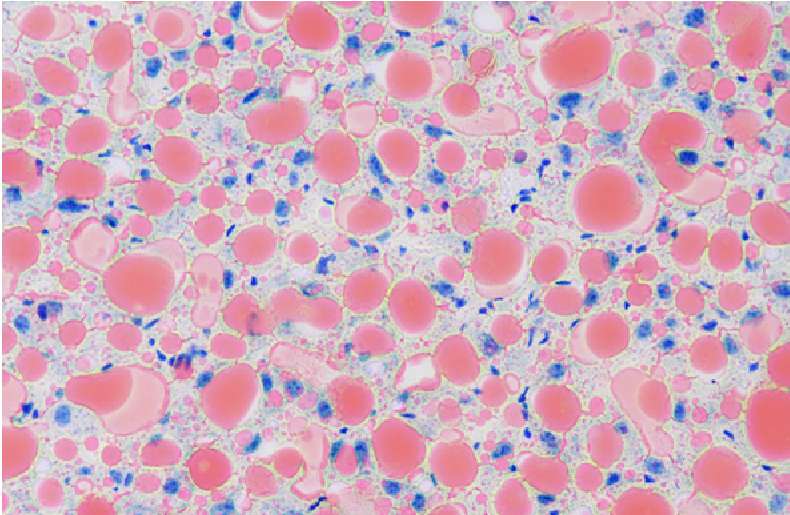
Staining of mouse liver sections showing steatosis of the liver (fatty liver), with accumulation of fat, lipid droplets (in red), within cells. Cell nuclei stain blue. Credit: C. Maillo, IRB Barcelona
Without CPEB4, the endoplasmic reticulum is unable to activate the stress response, thus causing hepatocytes to accumulate the lipids produced by the fatty liver.
New treatments?
Raúl Méndez, ICREA researcher at IRB Barcelona and co-leader of the study, explains that "knowledge of the hepatic function of CPEB4 could be useful as a predictive marker for those people with variants of this protein, thus serving to prevent this condition, for example, through improvements in diet and better choice of eating times. Such knowledge could also contribute to the development of treatments that boost the clean-up process".
The researchers have managed to reverse fatty liver disease in mice by treatment with a drug called Tudca, which is currently used for other disorders. This drug exerts the same function as the proteins that are activated by CPEB4 and that are responsible for cleaning up the cell, namely chaperones. "In the future, it may be possible to design molecules like Tudca that specifically target CPEB4, thus enhancing the liver clean-up process," says Méndez.
"This basic research study does not have a direct and immediate clinical application, but it lays down the foundation for the applied science that follows," says Mercedes Fernández, co-leader of the study and head of the group at IDIBAPS and the Biomedical Research Networking Center of Hepatic and Digestive Diseases (CIBEREHD).
Fernández warns, "Given the obesity epidemic in the U.S. and worldwide, an increase in those affected by non-alcoholic fatty liver disease is expected in the coming decades and we still do not have a suitable treatment for this condition; A fundamental understanding of this medical problem is therefore essential for development of novel treatment strategies."
It is estimated that between 80 and 100 million people in the U.S. alone suffer from fatty liver disease. People with this disease have an increased risk of cirrhosis and liver cancer. Moreover, liver cancer incidence has more than tripled since 1980 and is the primary cause of death in patients with cirrhosis.
More information: Carlos Maillo et al. Circadian- and UPR-dependent control of CPEB4 mediates a translational response to counteract hepatic steatosis under ER stress, Nature Cell Biology (2017). DOI: 10.1038/ncb3461
Journal information: Nature Cell Biology