Commercial weight-loss drug could help treat opioid addiction
Scientists are working to come up with new therapies to curb America's opioid epidemic and aid hospitals, doctors and public health officials in this fight. Now one team reports in the journal ACS Chemical Neuroscience that the commercial weight-loss drug lorcaserin, when given to rats who had been self-administering oxycodone, appeared to reduce their use of and craving for the opioid.
The U.S. Centers for Disease Control and Prevention estimates that the number of deaths in the U.S. from prescription opioid overdoses has quadrupled since 1999 to the present day. Existing treatments to help people quit using the drugs typically work by blocking or competing for the receptors that opioids bind to, dampening their euphoric effects. But people who have tried these medications often relapse, particularly in environments they associate with past drug use. Kathryn A. Cunningham and colleagues wanted to see if lorcaserin could help prevent this effect. Previous preclinical studies suggested that the medicine can curb cocaine and nicotine cravings, even when tested in tempting environments.
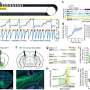
The researchers administered lorcaserin to rats who had been taking oxycodone. These rats were subsequently less likely to take oxycodone and exhibit drug-seeking behaviors than the rats that didn't get the treatment. This was true even when the lorcaserin-treated rats were exposed to cues such as lights and sounds that the animals were conditioned to associate with oxycodone.
More information: Harshini Neelakantan et al. Lorcaserin suppresses oxycodone self-administration and relapse vulnerability in rats, ACS Chemical Neuroscience (2017). DOI: 10.1021/acschemneuro.6b00413
Abstract
Opioid use disorder (OUD) is a major public health problem. High relapse rates and poor treatment retention continue to pose major challenges in OUD treatment. Of the abused opioids, oxycodone is well described to maintain self-administration and evoke the durable conditioned responses ("cue reactivity") that result from pairing of opioid-related stimuli (e.g., paraphernalia) with repeated abuse. Serotonin (5-HT) neurotransmission, particularly through the 5-HT2C receptor (5-HT2CR), regulates psychostimulant reward and cue reactivity, and in the present experiments, we investigated the hypothesis that the selective 5-HT2CR agonist lorcaserin, which is FDA-approved for the treatment of obesity, will suppress oxycodone self-administration and oxycodone-associated cue reactivity in rats. We found that lorcaserin inhibited oxycodone intake, an effect blocked by the selective 5-HT2CR antagonist SB242084. Lorcaserin also decreased responding for the discrete cue complex ("cue reactivity") previously associated with delivery of oxycodone (i.e., stimulus lights, infusion pump sounds) in both abstinence and extinction-reinstatement models. The selected dose range of lorcaserin (0.25-1 mg/kg) does not overtly alter spontaneous behaviors nor operant responding on inactive levers in the present study. Taken together, the ability of lorcaserin to reduce the oxycodone self-administration and decrease cue reactivity associated with relapse highlights the therapeutic potential for lorcaserin in the treatment of OUD.