Enteral DHA doesn't cut preemie bronchopulmonary dysplasia risk
(HealthDay)—Enteral docosahexaenoic acid (DHA) supplementation does not reduce the risk of bronchopulmonary dysplasia for infants born before 29 weeks of gestation, according to a study published in the March 30 issue of the New England Journal of Medicine.
Carmel T. Collins, Ph.D., from the University of Adelaide in Australia, and colleagues randomized 1,273 infants born before 29 weeks of gestation within three days after their first enteral feeding to receive an enteral emulsion providing DHA or a control emulsion without DHA until 36 weeks of postmenstrual age. Overall, 1,205 infants survived to the primary outcome assessment.
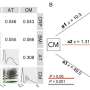
The researchers found that 49.1 percent of the 592 infants assigned to the DHA group and 43.9 percent of the 613 assigned to the control group were classified as having physiological bronchopulmonary dysplasia (relative risk adjusted for randomization strata, 1.13). The composite outcome of physiological bronchopulmonary dysplasia or death before 36 weeks of postmenstrual age occurred in 52.3 and 46.4 percent of the infants in the DHA and control groups, respectively (adjusted relative risk, 1.11).
"Enteral DHA supplementation at a dose of 60 mg per kilogram per day did not result in a lower risk of physiological bronchopulmonary dysplasia than a control emulsion among preterm infants born before 29 weeks of gestation and may have resulted in a greater risk," the authors write.
More information: Abstract/Full Text (subscription or payment may be required)
Copyright © 2017 HealthDay. All rights reserved.