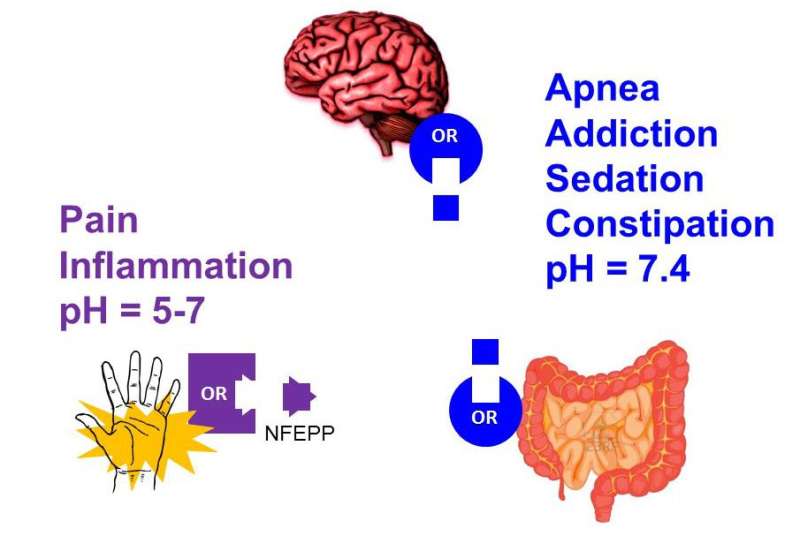
NFEPP activates opioid receptors in injured, but not normal, tissue. Credit: G C. Stein
(Medical Xpress)—A team of researchers with the Free University of Berlin and Zuse-Institut Berlin has developed a type of opioid that was shown to target pain in rats without causing negative side effects. In their paper published in the journal Science, the team describes the new opioid, how well it worked in rats and the side effects that were eliminated.
Most everyone knows about the positive and negative attributes of opioids—they are used to dull pain but are also highly addictive and have side effects such as constipation and respiratory distress. In this new effort, the researchers have developed a type of opioid they have named NFEPP that is works only on the part of the body that is in pain, while not affecting other parts, thus averting side effects.
As part of their research, the group noted that the parts of the body that hurt typically result in inflammation as the body tries to repair itself. They further noted that body parts experiencing inflammation tend to be more acidic than other parts. Because of that, they set about developing an opioid that would bind only to nerve receptors that exist in acidic environments. Opioids binding to untargeted parts of the body are what cause side effects, after all—binding to nerve cells in the gastrointestinal tract, for example, causes constipation and binding to nerve cells in the brain is what leads to feelings of euphoria and addiction.
Molecular dynamics simulation of opioid receptor in complex with NFEPP at low pH. Credit: V. Durmaz
The researchers tested the new opioid on rats, and found that it was comparable to the commonly prescribed opioid fentanyl in relieving pain, but it did not cause constipation, addiction, breathing problems, heart rate increases or changes to blood oxygen levels. In short, it appeared the new opioid alleviated pain as well as current opioids, but did not cause any noticeable side effects.
More research is required to test both the efficacy and safety of the opioid in rats and other test animals before it can be tested in humans, but if the opioid turns out to work in humans the way it does in rats, it could mark a truly transformative moment in medical science—a true breakthrough in pain mitigation and management.
Credit: G. Del Vecchio & V. Spahn
More information: V. Spahn et al. A nontoxic pain killer designed by modeling of pathological receptor conformations, Science (2017). DOI: 10.1126/science.aai8636
Abstract
Indiscriminate activation of opioid receptors provides pain relief but also severe central and intestinal side effects. We hypothesized that exploiting pathological (rather than physiological) conformation dynamics of opioid receptor-ligand interactions might yield ligands without adverse actions. By computer simulations at low pH, a hallmark of injured tissue, we designed an agonist that, because of its low acid dissociation constant, selectively activates peripheral μ-opioid receptors at the source of pain generation. Unlike the conventional opioid fentanyl, this agonist showed pH-sensitive binding, heterotrimeric guanine nucleotide–binding protein (G protein) subunit dissociation by fluorescence resonance energy transfer, and adenosine 3′,5′-monophosphate inhibition in vitro. It produced injury-restricted analgesia in rats with different types of inflammatory pain without exhibiting respiratory depression, sedation, constipation, or addiction potential.
Journal information: Science
© 2017 Medical Xpress