Antibiotic of last resort re-engineered to kill resistant bacteria
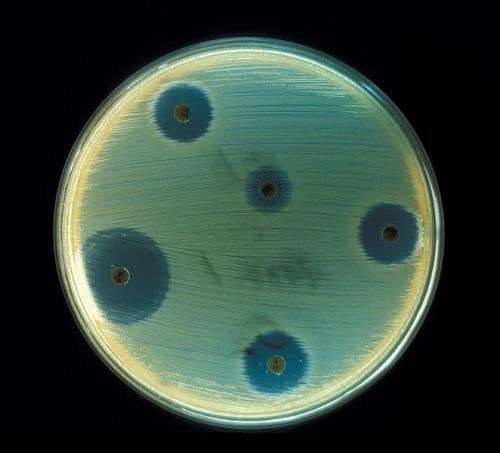
The frightening spread of antibiotic-resistant superbugs threatens to return medicine to the pre-antibiotic era, with the return of deadly infectious diseases long thought vanquished.
Each year, more than 2 million people in the United States get antibiotic-resistant infections, according to the Centers for Disease Control and Prevention. At least 23,000 of them die. Unless breakthroughs are achieved, that toll will keep rising.
If a new version of an antibiotic of last resort lives up to its promise, that date with doom may be averted. A study on this bolstered form of vancomycin by scientists at the Scripps Research Institute was released Monday.
Researchers led by Dale Boger, co-chair of Scripps' Department of Chemistry, introduced three modifications to vancomycin, all lethal to bacteria and independent of each other. Superbugs need to overcome all three changes to survive, which is extremely unlikely, the study said.
The study was published in the Proceedings of the National Academy of Sciences. Boger was senior author and Akinori Okano, also of Scripps, was first author. Research funding was supplied by the National Institutes of Health.
While fear of losing antibiotics has made doctors restrict their use, Boger and colleagues contend that doesn't solve the problem, it just slows the process. Better science is the solution, they say, to engineer antibiotics to anticipate and thwart resistance from the start.
The modified vancomycin needs further development so it can be tested in people to prove its safety and efficacy. Moreover, Boger's team made the drug through a lengthy, 30-step process, which limits yield. He and colleagues are working on shortening that process.
Even with the current synthesis method, the new vancomycin should be medicinally useful, the study said. It's more than 10 times as potent as an earlier version that introduced two lethal modifications. With the third lethal change, the altered vacomycin is more than 1,000 times as potent as standard vancomycin.
Two scientists praised the study as chemical synthesis of the highest order.
Medicinal chemist Derek Lowe said he's excited about the report and looking forward to seeing the new form of vancomycin developed further.
In a 2015 post on his blog, In the Pipeline, Lowe praised a previous study on the research that introduced the two lethal modifications.
That achievement arose from Boger's work in total synthesis of complex organic molecules. It's one of his specialties, and also something other Scripps scientists have been noted for.
Boger's previous work on vancomycin is the kind of feat that requires intense human creativity, something that can't be captured in automated synthesis, Lowe wrote in 2015.
The new study adds even more chemical tricks, he said.
"Vancomycin's original odd mechanism of action is why it's lasted so long in the first place. It doesn't bind directly to a bacterial protein, which would engage the evolutionary battle directly, but instead vacuums up a key oligopeptide that the bacteria need to function," Lowe said in an email.
"Throw these new functions in on top of that, and you not only get a big boost in potency, which this (latest) paper demonstrates, but you make it far, far less likely that a bacterium will emerge that will be able to deal with all of this at once."
Dan Kahne, a Harvard University professor of chemistry and chemical biology, described Boger's work as "spectacular."
"To be able to engineer a molecule of the complexity of vancomycin is a feat that few synthetic groups in the world could even attempt," Kahne said. "We need to continue to support these kinds of research programs because resistance will develop to any antibiotic, because death is a powerful selection."
Bacteria exist in astronomical numbers, mutate often and freely swap genes with others, even between species. So when they're confronted with antibiotics, Darwinian evolution by natural selection ensures that when resistance occurs even once, it rapidly spreads.
Vancomycin foiled this evolution for decades because it indirectly attacks bacteria by removing oligopeptide, a substance needed for their survival. But even so, some bacteria have developed resistance.
This process has spurred the rise of vancomycin-resistant enterococci, or VRE, three letters that strike fear into hospital staffs the world over. VRE has spread relentlessly, despite the best efforts of medical professionals.
Given the power of natural selection, the emergence of VRE and other resistant organisms appears inevitable. So doctors have become more cautious in prescribing antibiotics, hoping to reduce the pressures of natural selection that fuel resistance.
Boger and colleagues say in the study that approach is too defeatist.
"Although sounding attractive, the effort to restrict antibiotic use seems counter to their importance, introduces guilt into even their most legitimate of uses, challenges the prevailing practices of initial empirical best guess therapy and prophylaxis deployment, and produces additional disincentives to antibiotic development," the study says.
Moreover, restricting antibiotic use doesn't do anything to generate greater understanding of how resistance arises and how it might be thwarted, it said.
And the study posed a question:
"As an alternative to championing the restricted use of antibiotics or conceding that bacteria will always outsmart us, can durable antibiotics be developed that are capable of continued or even more widespread use?"
The modified vancomycin provides a positive answer, the authors say, not only by itself, but also as an approach to generating other more durable antibiotics.
More information: Akinori Okano el al., "Peripheral modifications of [Ψ[CH2NH]Tpg4]vancomycin with added synergistic mechanisms of action provide durable and potent antibiotics," PNAS (2017). www.pnas.org/cgi/doi/10.1073/pnas.1704125114
©2017 The San Diego Union-Tribune
Distributed by Tribune Content Agency, LLC.