Engineering human stem cells to model the kidney's filtration barrier on a chip
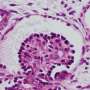
The kidney - made up of about a million tiny units that work to filter blood, constantly rids the body of undesired waste products to form urine while holding back blood cells and valuable proteins, and controls the body's fluid content. Key to each of these units is a structure known the 'glomerulus', in which so-called podocyte cells wrap themselves tightly around a tuft of capillaries separated from them only by a thin membrane composed of extracellular matrix, and leaving slits between them to build an actual filtration barrier. Podocytes are also the target of many congenital or acquired kidney diseases, and they are often harmed by drugs.
To build an in vitro model of the human glomerulus that could allow probing deeper into its function, as well as its vulnerabilities to disease and drug toxicities, researchers have been attempting to engineer human stem cells—that in theory can give rise to any mature cell type—so that they form into functional podocytes. These cell culture efforts, however, so far have failed to produce populations of mature podocytes pure enough as to be useful for modeling glomerular filtration.
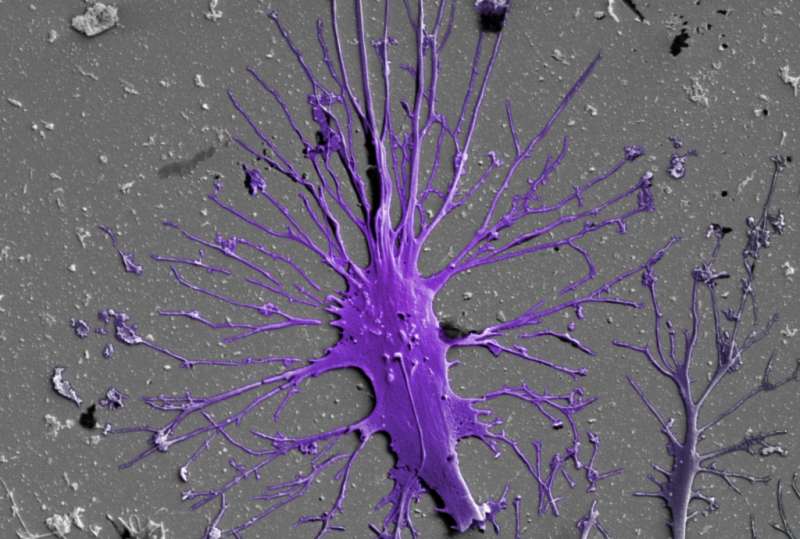
A team led by Donald Ingber, M.D., Ph.D., at Harvard's Wyss Institute of Biologically Inspired Engineering now reports a solution to this challenge in Nature Biomedical Engineering, which enables the differentiation of human induced pluripotent stem (iPS) cells into mature podocytes with more than 90% efficiency. Linking the differentiation process with organ-on-a-chip technology pioneered by his team, the researchers went on to engineer the first in vitro model of the human glomerulus, demonstrating effective and selective filtration of blood proteins and podocyte toxicity induced by a chemotherapy drug in vitro. Ingber is the Wyss Institute's Founding Director, the Judah Folkman Professor of Vascular Biology at Harvard Medical School (HMS) and the Vascular Biology Program at Boston Children's Hospital, as well as Professor of Bioengineering at the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS).
"The development of a functional human kidney glomerulus chip opens up an entire new experimental path to investigate kidney biology, carry out highly personalized modeling of kidney diseases and drug toxicities, and the stem cell-derived kidney podocytes we developed could even offer a new injectable cell therapy approach for regenerative medicine in patients with life-threatening glomerulopathies in the future," said Ingber.
Ingber's team has engineered multiple organs-on-chips that accurately mimic human tissue and organ-level physiology and that are currently being evaluated by the Food and Drug Administration (FDA) as a tool to more effectively study the effects of potential chemical and biological hazards found in foods, cosmetics or dietary supplements than existing culture systems or animal models. In 2013, his team developed an organ-on-a-chip microfluidic culture device that modeled the human kidney's proximal tubule, which is anatomically connected to the glomerulus and salvages ions from urinary fluid. Now, with the team's newly engineered human kidney glomerulus-on-a-chip, researchers also can get in vitro access to the kidney's core filtration mechanisms that are critical for drug clearance and pharmacokinetics, in addition to studying human podocytes at work.
To generate almost pure populations of human podocytes in cell culture, Samira Musah, Ph.D., the study's first author and HMS Dean's Postdoctoral Fellow who is working with Ingber at the Wyss Institute, leveraged pieces of the stem cell biologists' arsenal, and merged them with snippets taken from Ingber's past research on how cells in the body respond to adhesive factors and physical forces in their tissue environments.
"Our method not only uses soluble factors that guide kidney development in the embryo, but, by growing and differentiating stem cells on extracellular matrix components that are also contained in the membrane separating the glomerular blood and urinary systems, we more closely mimic the natural environment in which podocytes are induced and mature," said Musah. "We even succeeded in inducing much of this differentiation process within a channel of the microfluidic chip, where by applying cyclical motions that mimic the rhythmic deformations living glomeruli experience due to pressure pulses generated by each heart beat, we achieve even greater maturation efficiencies."
The complete microfluidic system closely resembles a living, three-dimensional cross-section of the human glomerular wall. It consists of an optically clear, flexible, polymeric material the size of a computer memory chip in which two closely opposed microchannels are separated by a porous, extracellular matrix-coated membrane that corresponds to the kidney's glomerular basement membrane. In one of the membrane-facing channels, the researchers grow glomerular endothelial cells to mimic the blood microvessel compartment of glomeruli. The iPS cells are cultured on the opposite side of the membrane in the other channel that represents the glomerulus' urinary compartment, where they are induced to form a layer of mature podocytes that extend long cellular processes through the pores in the membrane and contact the underlying endothelial cells. In addition, the device's channels are rhythmically stretched and relaxed at a rate of one heart beat per second by applying cyclic suction to hollow chambers placed on either side of the cell-lined microchannels to mimic physiological deformations of the glomerular wall.
"This in vitro system allows us to effectively recapitulate the filtration of small substances contained in blood into the urinary compartment while retaining large proteins in the blood compartment just like in our bodies, and we can visualize and monitor the damage inflicted by drugs that cause break-down of the filtration barrier in the kidney," said Musah.
More information: Samira Musah et al. Mature induced-pluripotent-stem-cell-derived human podocytes reconstitute kidney glomerular-capillary-wall function on a chip, Nature Biomedical Engineering (2017). DOI: 10.1038/s41551-017-0069