July 6, 2017 report
Neoadjuvant chemotherapy induces breast cancer metastasis through a TMEM-mediated mechanism
(Medical Xpress)—A team of researchers working at the Albert Einstein College of Medicine in the U.S. has found evidence that suggests administering chemotherapy to breast cancer patients prior to surgery can put them at increased risk for metastasis. In their paper published in the journal Science Translational Medicine, the group describes how they followed the treatment of 20 cancer patients and tested them for structures in the blood that indicate a higher risk for cancer spreading and what they found.
One of the most common ways to treat breast cancer is to administer chemotherapy and then to perform surgery to remove the tumor. It is believed that in addition to reducing tumor size, the chemotherapy drugs reduce the chance of the cancer spreading to other parts of the body. But now, it appears that that the latter assumption may not only be wrong, but the opposite of what actually occurs.
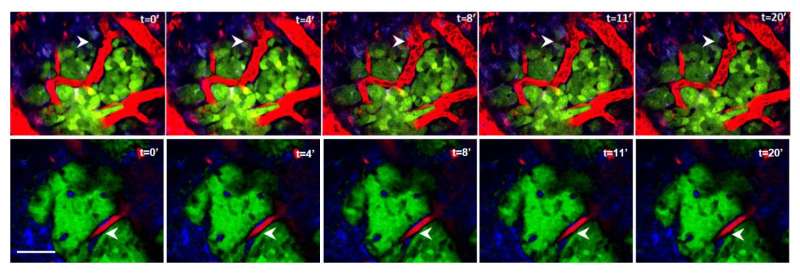
In this new effort, the researchers began by noting that some recent studies have suggested that instead of reducing the spread of cancer, some chemotherapy drugs might actually be making it easier for cancer cells to migrate to other body parts. To find out if that might be true, the team measured levels of tumor microenvironment of metastasis (TMEMs) in 20 breast cancer patients throughout their treatment. TMEMs are structures shown to be an indicator of metastasis. The researchers report that they found elevated levels of TMEMs in all of the patients that had received chemotherapy prior to having surgery.
The researchers also conducted research a breast cancer mouse model—they report that they found an increase in TMEM sites in mice that had received chemotherapy compared to those that did not.
The researchers also found that one such chemotherapy drug in particular, paclitaxel, caused bone marrow cells with TIE2 receptors to move to tumors, which resulted in TMEM formation. When they blocked the TIE2 with drugs, the number of TMEM sites in chemotherapy-treated mice did not increase.
The researchers suggest that more research needs to be done to better ascertain if chemotherapy drugs are causing cancers to spread and that in the meantime, some doctors may consider performing surgery first, then prescribing chemotherapy for breast cancer patients.
More information: Neoadjuvant chemotherapy induces breast cancer metastasis through a TMEM-mediated mechanism, Science Translational Medicine 05 Jul 2017: Vol. 9, Issue 397, eaan0026 , DOI: 10.1126/scitranslmed.aan0026 , stm.sciencemag.org/content/9/397/eaan0026
Abstract
Breast cancer cells disseminate through TIE2/MENACalc/MENAINV-dependent cancer cell intravasation sites, called tumor microenvironment of metastasis (TMEM), which are clinically validated as prognostic markers of metastasis in breast cancer patients. Using fixed tissue and intravital imaging of a PyMT murine model and patient-derived xenografts, we show that chemotherapy increases the density and activity of TMEM sites and Mena expression and promotes distant metastasis. Moreover, in the residual breast cancers of patients treated with neoadjuvant paclitaxel after doxorubicin plus cyclophosphamide, TMEM score and its mechanistically connected MENAINV isoform expression pattern were both increased, suggesting that chemotherapy, despite decreasing tumor size, increases the risk of metastatic dissemination. Chemotherapy-induced TMEM activity and cancer cell dissemination were reversed by either administration of the TIE2 inhibitor rebastinib or knockdown of the MENA gene. Our results indicate that TMEM score increases and MENA isoform expression pattern changes with chemotherapy and can be used in predicting prometastatic changes in response to chemotherapy. Furthermore, inhibitors of TMEM function may improve clinical benefits of chemotherapy in the neoadjuvant setting or in metastatic disease.
© 2017 Medical Xpress

















