Singapore scientists pave way for better juvenile arthritis diagnosis and treatment outcome prediction
A team of scientists and doctors from the SingHealth Duke-NUS Academic Medical Centre (AMC) has uncovered a new group of regulatory T (Treg) cells and DNA features associated with juvenile idiopathic arthritis (JIA), the most common form of arthritis among children under the age of 16. Their findings could potentially enhance diagnosis of the disease and prediction of therapy outcomes for improved treatment successes.
Results of their studies were published in the Annals of Rheumatic Diseases and Proceedings of the National Academy of Sciences.
JIA is a disease of the immune system that causes inflammation leading to pain, stiffness and swelling in patients' joints. It affects around one in 1,000 children in the world.
Juvenile arthritis has no cure and young patients can only alleviate pain or prevent joint deterioration through use of medication or therapy. In advancing care for JIA, researchers are keen to identify the culprit cells or genetic signatures behind the disease in order to tackle it.
1st discovery: Inflammation-associated Treg Cells
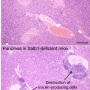
In its first discovery, the SingHealth Duke-NUS AMC research team identified a previously unknown group of Treg cells that is associated with inflammation in JIA. Treg cells are a subset of white blood cells that regulate the body's immune system. When the body has an imbalanced number of Treg cells, its immune tolerance can fail and experience autoimmune disorders such as arthritis.
The team found that the identified Treg cells play a role in JIA progression. During the active disease stage of JIA, these cells expand, grow in number, re-circulate through inflamed areas of patients' body, and migrate to the connective tissue of patients' joints. Additionally, a larger quantity of these cells can be found in JIA patients who cannot control arthritis inflammation and are unresponsive to therapy as compared to those who are.
"Clinicians could potentially use this novel group of cells as a marker to diagnose JIA in patients, as well as to predict or monitor patients' responsiveness to therapy. Importantly, these cells are readily detectable in patients' bloodstream, allowing for any clinical tests to be minimally invasive and pain-free for patients," said Professor Salvatore Albani, Director, SingHealth Translational Immunology and Inflammation Centre (STIIC), Professor, Duke-NUS Medical School and Senior Clinician Scientist, KK Women's and Children's Hospital (KKH), who is the principal investigator of the study.
2nd discovery: Patients' DNA affects JIA treatment outcomes
Currently, only about one-third of JIA patients get better after medication or therapy, while the rest continue to see their condition flare up even after treatment.
To accurately predict treatment outcomes, the research team studied JIA patients' treatment responses and found that epigenetics - or individuals' DNA and the way each body uses its genes - determined one's clinical "fate". In other words, the key is not in individuals' genetic make-up but rather, in how their bodies employ genes. Even patients with identical genetic backgrounds could experience different clinical outcomes based on their DNA features that activate genes differently.
One of the research paper's co-author, Associate Professor Thaschawee Arkachaisri, Head & Senior Consultant, Rheumatology and Immunology Service, KKH and Associate Professor, Duke-NUS, said "These discoveries could enable doctors to predict treatment responses and personalise treatment for patients. This is especially relevant for difficult JIA cases which may require more complex therapies, and is important to help save time and money, prevent treatment complications and ultimately, improve care outcomes."
The team's findings are also relevant for adult rheumatoid arthritis, a similar autoimmune condition that affects one in 100 adults in the world.