Credit: CC0 Public Domain
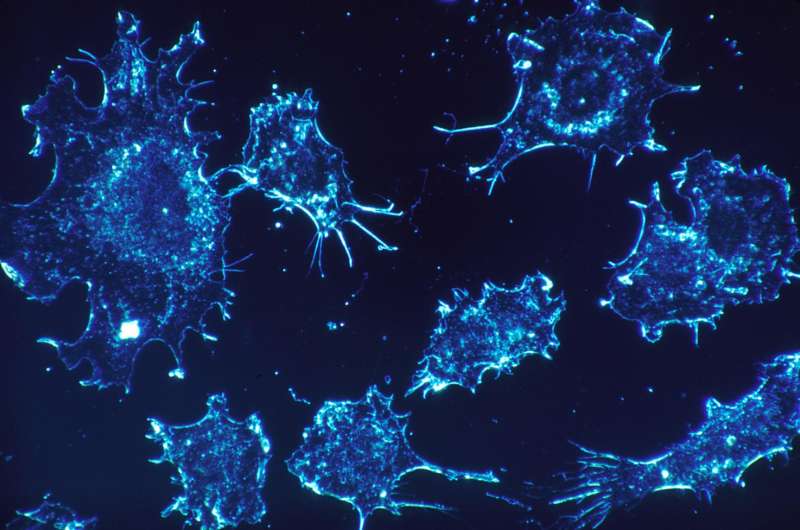
Heat therapy may be a promising treatment in the fight against cancer, autoimmune problems and efforts to avoid organ rejection in transplant patients, according to researchers at the University of Kentucky. The research team exposed colorectal cancer cells and T-cells—immune cells that fight disease—to temperatures lower (hypothermic) and higher (hyperthermic) than normal body temperatures. They observed the effects of these temperature changes on the energy production (bioenergetics) in both cell types. They will present their findings at the American Physiological Society's Physiological Bioenergetics: Mitochondria from Bench to Bedside conference in San Diego.
The use of thermotherapy to combat disease has a long history dating back to ancient Egypt. But much of what is known about heat therapy as a treatment for disease has been discovered through trial and error. With new bioenergetic measurement technologies such as the Seahorse XF analyzer, researchers are now able to apply more detailed, real-time, hypothesis-driven methods to research the effects of temperature on diseases.
"It is apparent that high temperature is naturally involved in immune response. As we get sick we normally develop some level of fever, which is involved with the body's normal response to infections. This activates the immune system to attack or eliminate infections," said Mihail (Mike) Mitov, PhD, lead author on the abstract. "A popular theory in the field of thermotherapy is that prolonged hyperthermia causes cell-cycle arrest in cancer cells. This explains some of the cancer-stopping effects of thermotherapy. But apparently, there are also changes in the energy metabolism of cells and possibly other yet unknown processes that affect the energetic profiles of the cells. These processes could be exploited for the development of new treatments or therapeutic approaches with fewer side effects than chemotherapy."
Normal function of immune cells, such as T-cells, rely heavily on an activation process involving increased glycolysis—the breakdown of glucose into pyruvate. Such switches in T-cell metabolism are believed to help boost the cells' ability to fight off infection and disease. Cancer cells are also known to be more glycolytic compared to normal non-cancer cells. "The present study investigates how changes in temperature affect the bioenergetics characteristics (mitochondrial function and glycolysis) of three colorectal cancer cell lines and primary human T-cells utilizing Seahorse XF96 technology," the research team wrote.
The researchers performed experiments on the cells at 32, 37 and 42 degrees Celsius and found that temperature significantly changed multiple components of the glycolytic process in all cell types. "We found that short-term hyperthermia (a 2.5-hour exposure) increases glycolysis of T-cells, thus, putting them in an active state that potentially is ready to fight cancer or infections," Mitov noted. However, if the exposure to hyperthermia is prolonged the T-cells become inactive, which could be beneficial for people who need a suppressed immune response, such as those with an organ transplant or autoimmune diseases.
"These findings provide valuable insights into the metabolic and bioenergetics changes of colorectal cancer cells and human T-cells under hypo- and hyperthermia conditions that could potentially lead to the development of better-targeted and personalized strategies for patients with cancer, metabolic disorders, diabetes or transplanted organs," the research team wrote.
Provided by American Physiological Society