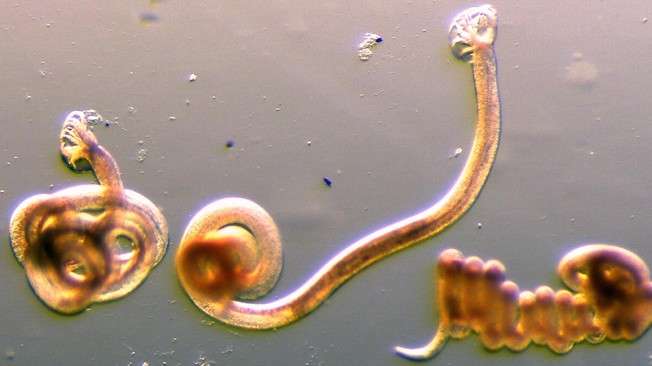
The nematode Heligmosomoides Polygyrus, used as a gastrointestinal parasitic model to understand the mechanism behind lymph node lymphangiogenesis. Credit: Lalit Kumar Dubey/EPFL
By studying a worm infection, EPFL scientists have discovered how lymphatic vessels grow within lymph nodes, with major implications for cancer and inflammation.
Lymph nodes are small, kidney-shaped organs found throughout the body. Full of immune cells, their function is to clear out foreign objects and support the immune system. Lymph nodes communicate with the tissues and with each other through the lymphatic vessels, which carry fluids and objects from the tissues and back out to the bloodstream. Normally, lymphatic vessels grow during the embryo stage, but also in adults during wound healing, cancer, and inflammation. But the exact mechanism of this "lymphangiogenesis" is yet unknown. EPFL scientists have now identified the molecules that signal the growth of lymphatic vessels during worm infections. The work is published in Nature Communications.
The lymphatic vessels drain pathogens from tissues to the collecting lymph nodes, where immune responses begin. They also allow lymphocytes and dendritic cells —which expose pathogen material to trigger the immune system—to flow in and out of the lymph nodes. Because of this, lymphangiogenesis is important for immune responses against infections.
But recent studies have shown that lymphangiogenesis can also regulate immune responses during inflammation. This connection between inflammation and lymphangiogenesis is key in our understanding of the adaptive immune response, which is the slower but more specialized wave against infections and involves T and B cells.
The lab of Nicola Harris at EPFL looked at the mesenteric lymph node, which collects fluids and objects from the intestine of mice. The research, led by Lalit Kumar Dubey, follows a 2016 paper from the group, which showed how worm infection stimulates B cells to 'talk' to the endothelial cells of the mesenteric lymph node and kickstart an immune response.
In the current study, the researchers used immunofluorescent imaging of whole tissues to generate a picture of how cells interact with each other when the mesenteric lymph node is infected with the worm Heligmosomoides polygyrus bakeri (Hpb), which is a standard tool in the field. They then determined the molecular nature and consequences of these interactions.
The imaging showed that Hpb infection causes extensive lymphangiogenesis inside the mesenteric lymph nodes of mice. This is also accompanied by an increased influx of dendritic cells, signifying the beginning of an immune response.
"We uncovered a multidirectional communication between B lymphocytes, fibroblasts, and lymphatic endothelial cells inside the lymph nodes," says Nicola Harris. "This three-way communication triggers lymphangiogenesis following intestinal infection with the Hpb worm."
Even more importantly, the scientists also identified the actual biological molecules involved in this process. These include both cell-bound and secreted proteins, which the three cell types use to communicate with each other.
Specifically, the lymphangiogenesis was driven by a complex interplay between inflammatory cytokines, fibroblastic reticular cells, and B cells in the lymph node. The end result is the release of a protein called B-cell activating factor, which in turn stimulates the production of vascular endothelial growth factor (VEGF) from B cells. The latter drives the proliferation of endothelial cells for the formation of new lymph vasculature.
The study demonstrates a novel, previously unknown dimension of lymphangiogenesis, involving cross-talk between cells of the mesenteric lymph node. The findings can help our understanding and perhaps future treatment of immune diseases and perhaps even some forms of cancer.
More information: Lalit Kumar Dubey et al. Interactions between fibroblastic reticular cells and B cells promote mesenteric lymph node lymphangiogenesis, Nature Communications (2017). DOI: 10.1038/s41467-017-00504-9
Journal information: Nature Communications
Provided by Ecole Polytechnique Federale de Lausanne