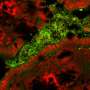
Gut microbes may influence multiple sclerosis progression
Researchers at UC San Francisco have identified specific gut microbes associated with multiple sclerosis (MS) in human patients, showing that these microbes take part in regulating immune responses in mouse models of the disease.
The new findings — to be published during the week of September 11, 2017 in the Online Early Edition of Proceedings of the National Academy of Sciences (PNAS)—suggest that gut microbes may play a role in the neurodegeneration that characterizes MS. The authors hope the finding will help scientists understand the origins of MS and potentially lead to treatments, such as dietary changes or drugs based on microbial byproducts, which could improve the course of the disease.
MS is an autoimmune neurodegenerative disorder that affects approximately 2.5 million people worldwide with progressive loss of vision, weakness and tremors, and problems with coordination and balance. In severe cases, paralysis may result.
MS is caused when the immune system attacks the insulation, known as myelin, that wraps around nerve cells. Researchers have learned a great deal about MS over the past decades, but continue to puzzle over why the immune system turns against myelin in the first place.
"The field has been very successful in identifying genes associated with susceptibility to MS, but I've never been satisfied with the amount of risk that we'll be able to explain with just genetics," said Sergio Baranzini, PhD, a professor of neurology at UCSF and the senior author on the new study. "Even identical twins, who share the same genetic inheritance, only share an MS diagnosis about 35 percent of the time. It's clear the genome is important, but environmental factors must also play a major role."
Many suspected environmental contributors to MS, such as history of smoking, diet, and environmental exposures, are very hard to pin down and associate with a biological impact on patients, Baranzini said.
But a growing number of studies demonstrating that gut microbes can directly influence the function of the human immune system suggested a new possibility to Baranzini: Since "the intestine is actually the most intimate connection between the outside world and the immune system," he explained, the human gut microbiome could play a role in the onset or progression of MS.
In the new study, postdoctoral researcher Egle Cekanaviciute, PhD, and collaborators analyzed the gut microbiome of 71 MS patients as well as 71 healthy control subjects, and identified specific species of bacteria that were either more or less common in people with multiple sclerosis than in the general population. Then they turned to the much more difficult task of investigating how these differences in gut bacteria might influence the immune system's attack on myelin in MS.
"A lot of microbiome studies say, 'These bacteria are increased in patients with a disease, and those bacteria are reduced.' And then they stop," Cekanaviciute said. "We wanted to know more: should we care about the ones that are increased because they are harmful or the ones that are decreased because perhaps they are helpful? What do these microbes actually do that could have an impact on people's health?"
First, the team explored whether components of these bacteria could alter the behavior of human immune cells to make them either pro- or anti-inflammatory. In laboratory dishes, the researchers exposed human immune cells to bacterial extracts, and found that two species that were more common in people with MS — Akkermansia muciniphila and Acinetobacter calcoaceticus—triggered the cells to become pro-inflammatory, while another found at lower than usual levels in MS patients — Parabacteroides distasonis—triggered immune-regulatory responses.
To see how these bacteria might affect the immune system as a whole, the researchers then introduced each of these three species into mice that otherwise lacked a microbiome and found that the bacteria had a similar effect: A. muciniphila and A. calcoaceticus triggered inflammatory immune responses, while P. distasonis tamped down inflammation.
But these experiments only examined the impact of one bacterial species at a time — the question remained how the complex microbial ecosystems of MS patients might impact neurodegeneration. To answer this question, the team performed fecal transplants on mice with an experimentally induced form of MS. The researchers found that replacing the microbiomes of these mice with the microbiomes of MS patients caused the animals to lose key immune-regulatory cells and to develop more serious neurodegeneration, suggesting that the microbiome alone could contribute to the progression of MS.
A second study published in the same issue of PNAS—led by researchers at the Max Planck Institute in Germany and co-authored by Cekanaviciute and Baranzini—also found that microbiome transplants from MS patients could exacerbate symptoms in mice with a genetic model of the disease.
"Two different groups, using two separate cohorts of patients and controls, and two distinct mouse models of the disease, saw very similar results," Cekanaviciute said. "This is very promising evidence that we're on the right track."
The new findings suggest that the microbiome may play a key role in the origins of MS. "To be clear, we don't think the microbiome is the only trigger of MS," Cekanaviciute said. "But it looks like these microbes could be making the disease progression worse or better—pushing someone with genetic predisposition across the threshold into disease or keeping them safe."
The authors hope that future research will shine light on exactly how these bacterial populations influence the development and progression of MS. For instance, the authors note that at least one of the MS-associated bacteria produce molecules that mimic proteins found in myelin, suggesting that the presence of these bacteria could confuse the immune system into attacking myelin as well as the bacteria.
The authors also note that P. distasonis, the bacterial species found at lower-than-normal levels in MS patients, is thought to help the immune system learn to control its response to non-threatening gut microbes. Lacking these bacteria may allow the immune system to overreact to harmless microbes in people with MS, leading to destructive inflammation.
Baranzini said he hopes the field will be able to use these findings to develop novel therapies for patients with MS. "The microbiome is very malleable," he said. "You could relatively easily change it in an adult who has MS or is susceptible - something you cannot do with their genetics. This is not a magical approach, but it is hopeful."
More information: Egle Cekanaviciute el al., "Gut bacteria from multiple sclerosis patients modulate human T cells and exacerbate symptoms in mouse models," PNAS (2017). www.pnas.org/cgi/doi/10.1073/pnas.1711235114
Kerstin Berer el al., "Gut microbiota from multiple sclerosis patients enables spontaneous autoimmune encephalomyelitis in mice," PNAS (2017). www.pnas.org/cgi/doi/10.1073/pnas.1711233114