Michael Peters, Ph.D. Credit: Virginia Commonwealth University
A "twist of fate" that is minuscule even on the molecular level may cause the development of Alzheimer's disease, VCU researchers have found.
Observations of molecules called monomers, which are present in the brain, have revealed valuable insights into the disease's pathway. Monomers resemble a chain in which each of the links is one of 20 amino acids, the building blocks of essential proteins. Problems arise when these monomer chains repeatedly stack on top of one another to form harmful atypical amyloid fibrils, which are long, string-like structures.
Typical amyloid fibrils are made from monomer chains that contain 40 amino acid links, and may have a role in DNA transcription and other cellular processes. A comprehensive look at the brains of familial (inherited) and senile (non-inherited) Alzheimer's sufferers shows that monomers with two additional links in the amino acid chain—called Aβ42 monomers—lead to the devastating disease, said Michael Peters, Ph.D., professor in the Department of Chemical and Life Science Engineering in the School of Engineering.
In cases of familial Alzheimer's, mutations in the monomer production pathway leads to the higher number of monomers. It is currently unknown how they develop in brains affected by senile Alzheimer's.
Peters said it's "very bizarre" that the addition of these two naturally occurring amino acids to the end of a monomer is so consequential.
"With diseases like cancer you can have one or more mutations or sequence changes along the chain itself, which radically alters the protein's behavior," Peters said, "In the case of Alzheimer's, there are no mutations in the monomers. You are simply adding only two natural amino acids to the very end of the non-toxic monomers, which causes a catastrophe."
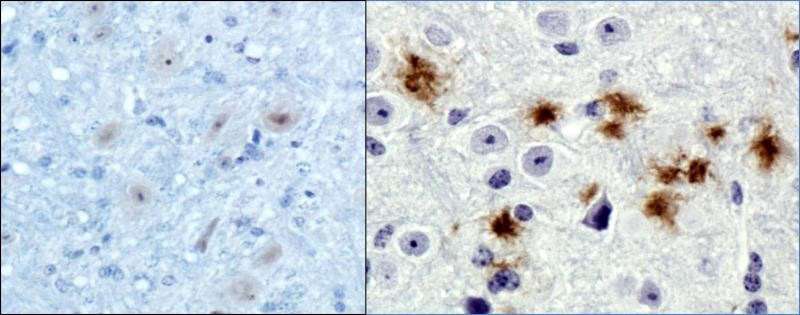
Left: Normal brain tissue. Right: Plaque stains from abnormal amyloid fibrils. Credit: Virginia Commonwealth University
A twist of fate
In a healthy brain, amyloid fibrils constructed from normal monomers in a specific sequence stack neatly on top of each other, similar to floors in a building. These fibril stacks weaken as they build and can break down and "fall over."
But this isn't the case for fibrils made from Aβ42 monomers, which are much stronger. Instead of breaking, these monomers continue to stack until they form long and resilient fibrils. These fibrils are believed to cause Alzheimer's disease because they invade the membranes of neurons, which leads to cell death.
Peters and School of Engineering Ph.D. candidate Oscar Bastidas conducted computational analysis to determine why fibrils constructed of Aβ42 monomers are so resilient. They determined the two additional amino acids at the monomer end caused slight rotations of molecules in specific locations within the fibrils. This allowed the monomers to stack slightly closer together, which contributed to resiliency.
"It's a literal twist of fate," Peters said.
New drugs
Peters and Bastidas are using this research to develop drugs to disrupt the formation of fibrils constructed from Aβ42 monomers without harming the brain.
"We have to get therapeutic molecules across the rather impervious blood-brain barrier and inhibit fibril formation, while not being toxic to the body or interfering with normal bodily functions," Peters said. "This makes for a very difficult three-fold problem, but we are making progress."
Provided by Virginia Commonwealth University