APC protein affects colon cancer immunity by preventing pre-cancerous inflammation
Adenomatous polyposis coli (APC) is a gene whose mutations are associated with a rare, hereditary form of colorectal cancer known as familial adenomatous polyposis. Research led by scientists at the Institut Pasteur and Inserm have recently demonstrated that mutations to this gene do not only lead to the emergence of colon polyps, they also harm the immune system, leaving it unable to tackle inflammation of the colonic mucosa. This dual impact supports the development of cancer. The finding, published in the journal Cell Reports on October 3rd, 2017, advances scientific knowledge on the development mechanisms of colorectal cancer.
Familial adenomatous polyposis is an inherited condition characterized by the formation of a very large number of polyps, small growths on the inner surface of the colon and the rectum which can develop into tumors beginning in adolescence. If left untreated, these polyps may result in colorectal cancer before the age of 40.
Colon cancer is one of the most deadly forms of cancer, and familial adenomatous polyposis currently represents 1 percent of all cases of colorectal cancer. Those affected by this hereditary disease therefore need close medical supervision.
Research led by scientists from the Institut Pasteur and Inserm recently demonstrated that mutations in the adenomatous polyposis coli (APC) gene, known to be involved in familial adenomatous polyposis, do not only lead to the emergence of colon polyps; they also harm the immune system, leaving it unable to tackle inflammation of the colonic mucosa. This dual impact may favor the development of cancer.
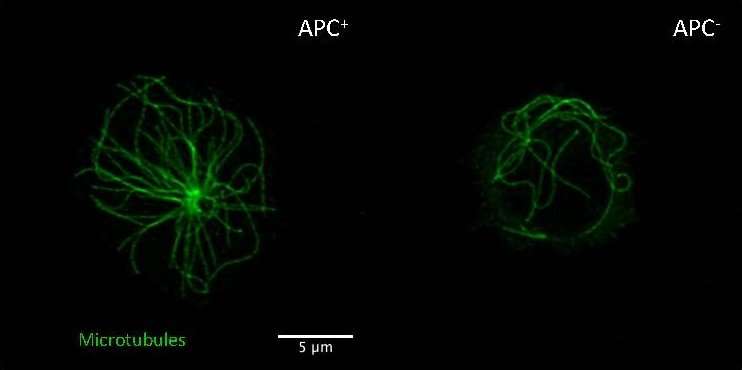
As Andrés Alcover, head of the Lymphocyte Cell Biology Unit at the Institut Pasteur and co-author of the paper, says, "The APC protein, associated with the microtubule cytoskeleton, has a major effect on the structure and differentiation of intestinal epithelial cells. By disrupting these functions in intestinal cells, APC mutations can lead to the development of tumors."
Scientists already knew that APC mutations could influence the immune system, but they had not yet identified the molecular mechanisms involved and the link with colorectal cancer development. The teams of scientists report that the APC protein activates T lymphocyte immune cells. "The protein activates T lymphocytes using a factor known as NFAT," says Alcover. "Polyposis patients have a mutant version of the gene, which leads to a deficiency in APC protein and could reduce the presence of NFAT in cell nuclei," thereby preventing lymphocyte activation.
One family of T lymphocytes, the regulatory T cells, present in large numbers in the intestine, is particularly sensitive to APC mutations. The scientists observed a dysfunction in these regulatory T cells in mice with these mutations that are predisposed to develop polyposis like the patients. This dysfunction leads to a deregulation of the immune system in the intestine and a failure to control local inflammation. "This is the first time that we have characterized at molecular level how mutations in the APC protein affect the immune system, creating favorable conditions for cancer development," says Alcover.
These findings suggest that mutations in the APC gene play a dual role in the development of colorectal cancer. Not only do they trigger the development of polyps, they also reduce the action of the immune system, preventing it from controlling gut inflammation. This vicious circle supports the development of cancer.
It remains to be seen whether defects in the APC protein in patients with familial adenomatous polyposis have consequences for the other cells in the immune system, especially those that directly eliminate cancer cells. If so, this research might pave the way for the development of new therapies to improve the efficacy of treatment for patients with familial adenomatous polyposis or other forms of intestinal cancer.
More information: Sonia Agüera-González et al, Adenomatous Polyposis Coli Defines Treg Differentiation and Anti-inflammatory Function through Microtubule-Mediated NFAT Localization, Cell Reports (2017). DOI: 10.1016/j.celrep.2017.09.020