The molecular mechanisms of Alzheimer's
Late-onset sporadic Alzheimer's disease (AD) accounts for about 99 percent of all cases, and is linked to multiple factors and pathogenetic mechanisms. One important contributor is a protein post-translational modification (PTM), known as O-linked β-N-acetylglucosamine, or O-GlcNAc.
Low levels and the labile nature of O-GlcNAc have complicated detailed molecular studies of this important post-translational modification at the level of the proteome. Scientists have also been unable to quantify O-GlcNAc on a large scale.
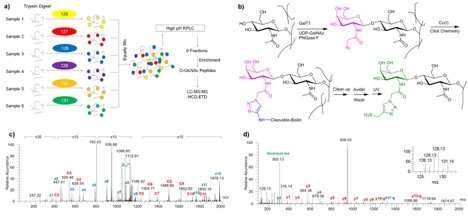
A recent paper in the Journal of Pathology reports on a new integrated proteomic pipeline that promises to harness advances in proteomics in ways that will ease investigations into hard-to-analyze O-GlcNAc. The study's authors are Sheng Wang, Tao Liu, eight other researchers at the Pacific Northwest National Laboratory (PNNL), and Cheng-Xin Gong of the City University of New York.
The study is the most comprehensive quantitative proteomic study of brain O-GlcNAcylation to date. Its use of multiplexed isobaric labeling with a pooled common reference strategy and an efficient and specific enrichment method enabled the comparison of more than a thousand O-GlcNAc peptides across many samples. That opens new avenues for future investigations of the molecular mechanisms of AD.
Moreover, the same quantitative proteomics analytical pipeline can be expanded for higher throughput and other forms of integrated proteomics PTM analysis (phosphorylation, for example) to reveal more about the relationship between O-GlcNAcylation and AD pathology.
Enter Advances in Proteomics
O-GlcNAc modifies many proteins and plays important roles in regulating diverse cellular processes, including metabolism, signaling, transcription, and protein quality control. It has recently attracted attention for its possible link to AD.
A comprehensive understanding of brain protein O-GlcNAcylation and its dysregulation in AD may contribute to a better understanding of the molecular mechanisms leading to AD.
Recent advances in proteomics technology make it possible to sidestep present analytical challenges to characterizing O-GlcNAc peptides. The PNNL-led paper records and demonstrates an integrated proteomic pipeline that harnesses some of these advances, combining isobaric tandem mass tag labeling with an efficient O-GlcNAc enrichment strategy.
The new analytical pipeline allowed researchers to characterize O-GlcNAcylation in postmortem human brains from 20 individuals - 10 with AD and 10 without AD or associated neurological pathologies or symptoms. Using frozen brain tissue samples, the PNNL-led team identified 1,850 O-GlcNAc peptides from 530 proteins, and confidently assigned more than half (1,094) to O-GlcNAc sites.
Among those O-GlcNAc proteins, 131 were altered significantly in brain tissue linked to AD, and covered a variety of functional categories, notably structural, synaptic, and memory-associated proteins.
Rapid, Reliable Comparisons
The new proteomic analysis pipeline enabled the researchers to rapidly and reliably compare the levels of more than 1,000 O-GlcNAc peptides, and to discover the AD-linked changes in more than 100.
This study is the most comprehensive quantitative proteomic study of brain O-GlcNAcylation to date. Future studies will feature more samples and integrated proteome-phosphoproteome-O-GlcNAc-ome analysis. In all, the aim is to shed light on AD pathology by listening in on the crosstalk between protein phosphorylation and O-GlcNAcylation.
The number of samples in the study is small, and all of the observed changes might not be solely due to AD. But the study's identification of altered O-GlcNAc peptides opens new avenues for future investigations of the molecular mechanisms of AD.
More information: Sheng Wang et al. Quantitative proteomics identifies altered O-GlcNAcylation of structural, synaptic and memory-associated proteins in Alzheimer's disease, The Journal of Pathology (2017). DOI: 10.1002/path.4929