Herpes virus mutant points towards new vaccine strategy
A Northwestern Medicine study published in PLOS Pathogens describes a newly-designed mutant herpes virus called R2 that provides a strategy for the development of novel herpes virus vaccines.
Herpes simplex viruses, known for causing cold sores and genital herpes, are two members of the alpha herpes virus family, which also includes the varicella-zoster virus, the cause of chickenpox. These viruses share a unique property: the ability to routinely and robustly infect the nervous system, establishing lifelong infection.
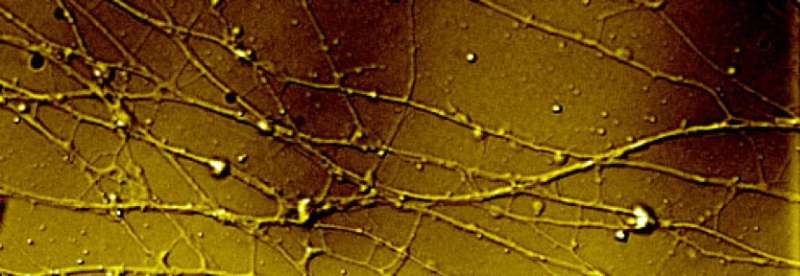
When a herpes virus initially enters a skin, or epithelial, cell, it begins shedding its outer layers, releasing viral proteins to perform their functions, but a small subset of viral proteins remains bound to the DNA-filled capsid, a protein shell that holds the DNA that will turn the cell into a herpes virus factory.
One of the proteins that escorts the DNA-filled capsid is the focus of the current study. Collaborators at Tufts University found that this protein, called protein UL37, has three distinct regions conserved in alpha herpes viruses, and the scientists discovered that mutating the second region, termed R2, exhibited an exciting trait.
Lead author Alexsia Richards, Ph.D., a former postdoctoral fellow in the laboratory of senior author Gregory Smith, Ph.D., professor of Microbiology-Immunology, explained that although the R2 mutant successfully infected epithelial cells, the virus failed to infect neurons.
The scientists discovered that R2 could enter neurons but once inside, it simply bounces back and forth, unable to travel the long distances needed to reach the nucleus of a neuronal cell where latent—or lifelong—infection is established.
To further understand these results, the scientists turned to mouse models. Using another alpha herpes virus that normally infects pigs, the scientists saw that mice infected with the unmodified version of the virus died rapidly, but all of the mice infected with the R2 mutant survived.
Next, the scientists investigated whether the R2 mutant can vaccinate mice. Collaborators at the University of Nebraska-Lincoln exposed mice to the R2 mutant, waited several days, and then infected the mice with 10,000 times the lethal dose normally required to kill 50 percent of the mice. They found that 75 percent of the vaccinated mice survived the lethal experiment with no signs of infection.
"We now have a virus with this unique property that had never been seen before: a virus that can propagate just like a wild-type herpes virus but specifically can't travel into the nerves and go latent in the nervous system," Smith said.
Furthermore, because mutating R2 produces an effect both in human and animal herpes viruses, the authors believe they've created a tool for a new generation of herpes virus vaccines.
"It's basically a tool to make any vaccine strain you want," Richards said.
The authors note that the R2 mutation findings have the potential to improve currently approved vaccines, like the chickenpox virus vaccine.
Future research will focus on further developing the R2 mutant as a master herpes virus vaccine.
"We are amazed by the efficacy of this vaccine, but this gets to the point where basically you need to partner with a pharmaceutical company. The hope would be that once the paper is out, and people start seeing the efficacy of this approach, they'll take interest and there'll be people that want to try and push this forward with our help," Smith said.
More information: Alexsia L. Richards et al. The pUL37 tegument protein guides alpha-herpesvirus retrograde axonal transport to promote neuroinvasion, PLOS Pathogens (2017). DOI: 10.1371/journal.ppat.1006741