ROS-scavenging nanozymes for anti-inflammation therapeutics
The dysregulation of reactive oxygen species (ROS) is linked to inflammatory diseases including rheumatoid arthritis, cardiovascular disease and cancer. Live organisms have therefore evolved a number of highly efficient anti-inflammation enzymes with ROS-scavenging capabilities to protect tissues from inflammation-induced damage. However, the natural ROS-scavenging enzymes are sensitive to environmental conditions and are hard to mass produce. To address these challenges, numerous artificial enzymes with ROS-scavenging capabilities have been developed. Among them, ROS-scavenging nanozymes have recently attracted great interest owing to their enhanced stability, multi-functionality and tunable activity.
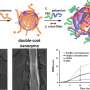
Nanozymes are catalytic nanomaterials with enzyme-mimicking activities. Several nanomaterials have been explored to develop ROS-scavenging nanozymes. For example, ceria nanoparticles (CeO2 NPs) have been demonstrated to possess superoxide dismutase (SOD)-mimicking activities due to the mixed valance states of Ce3+ and Ce4+. Biological studies have revealed that natural Mn SOD is superior to Cu/Zn SOD and Fe SOD, which implies that Mn-based nanozymes may have enhanced ROS-scavenging activities compared with known examples. Despite great promise, only a few Mn-based nanozymes have been reported. Moreover, they have not been used for in vivo anti-inflammation yet.
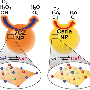
To tackle these challenges, Professor Wei at Nanjing University and his co-workers have now fabricated Mn3O4 NPs with multiple enzyme mimicking activities. The Mn3O4 nanozymes possessed SOD- and catalase-like activities as well as hydroxyl radical scavenging activity. Therefore, they scavenged the superoxide radical as well as hydrogen peroxide and the hydroxyl radical. Wei et al. also demonstrated that the Mn3O4 nanozymes were superior to CeO2 nanozymes in term of the ROS-scavenging activities. Moreover, they showed that the Mn3O4 nanozymes not only exhibited excellent ROS removal efficacy in vitro, but also effectively protected live mice from ROS-induced ear inflammation in vivo.
Their studies provided not only a highly efficient ROS-scavenging nanozyme, but also a promising therapeutic strategy for treating inflammation-related diseases.
More information: 10.1039/c7sc05476a Jia Yao et al. ROS scavenging Mn3O4 nanozymes for in vivo anti-inflammation, Chemical Science (2018). DOI: 10.1039/C7SC05476A