Investigators at Vanderbilt-Ingram Cancer Center (VICC) and the Virginia Tech Carilion Research Institute (VTCRI) have revealed a gene mutation's role in Von Hippel-Lindau syndrome, a genetically inherited disease which causes tumor growth in several organs.
The study led by corresponding authors W. Kimryn Rathmell, MD, Ph.D., director of the Division of Hematology and Oncology at VICC, and John Chappell, Ph.D., assistant professor at VTCRI's Center for Heart and Regenerative Medicine, was published recently in JCI Insight, a journal of the American Society for Clinical Investigation.
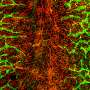
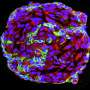
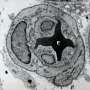
Von Hippel-Lindau syndrome (VHL) is a rare disease that causes benign tumors to grow in a variety of organs and can lead to cancer. The benign lesions in VHL are characterized by newly formed blood vessels that can cause serious or life-threatening complications especially if they occur in the retina, spinal cord, brain or inner ear.
"These growths have the most unusual vasculature I've ever seen," Chappell said. "It's chaotic and mispatterned, and as soon as I saw it, I knew I wanted to figure out how this mutation could lead to such disorganized vessels."
Rathmell and Chappell started investigating the gene mutation implicated in VHL while both worked at the University of North Carolina at Chapel Hill. Rathmell had developed a mouse model to study conditionally-induced gene mutations similar to the mutations that lead to VHL in humans.
"We have known for a long time that patients with Von-Hippel Lindau syndrome display variable features of the disease," Rathmell said. "These animal models were constructed to allow us to explore the range of effects caused by different types of mutations occurring in the same gene."
Rathmell was interested in how the mutated gene induced blood vessels to remodel into tumors especially since, when healthy, the gene helps suppress tumors. The team found that the mutation severely reduced the manner by which vessels branched out in later stages of development. It also significantly accelerated the maturation of arterial vessels, especially in the cells that line the blood vessels called pericytes. These cells wrap around the endothelial cells that line blood vessels, helping regulate blood flow and maintaining stability of the vessels.
The investigators conducted a large genetic screen and found that gene expression changed within several key signaling pathways, including the Notch pathway. Normally the Notch pathway helps cells communicate and regulates growth and differentiation of neurons and vascular cells during development. The VHL protein helps the body sense oxygen levels which influences other actions, including the pattern of vessel branching, in part through crosstalk with the Notch pathway.
The team found that the mutated gene confuses those signals, disrupting the oxygen-sensing abilities and influencing vessel growth.
"We suspected this change, but the extent of it was surprising," Chappell explained. "Von Hippel-Lindau mutations not only cause abnormal blood vessel growth, but they also cause new and existing vasculature to remodel in unusual ways."
The investigators believe this newly described pathway could be a target for potential therapies.
More information: Alexandra Arreola et al. Von Hippel-Lindau mutations disrupt vascular patterning and maturation via Notch, JCI Insight (2018). DOI: 10.1172/jci.insight.92193
Provided by Vanderbilt University