April 19, 2018 report
Mechanism that drives development of liver cancer brought on by non-alcoholic fatty liver disease discovered
A team of researchers from several institutions in China has found a mechanism that appears to drive the development of a type of liver cancer not caused by alcohol consumption. In their paper published in the journal Science Translational Medicine, the group describes their study of liver cancer in mouse models and what they found. Ngee Kiat Chua, Hudson Coates and Andrew Brown with the University of New South Wales offer a Focus piece on the work done by the team in China in the same journal issue.
Most everyone knows that alcoholics are at risk of developing liver cancer due to the impact of alcohol on their livers over the course of a lifetime. Less well understood are types of liver cancers that can be attributed to obesity-related factors (primarily because of high cholesterol levels) and more specifically, non-alcoholic fatty liver disease (NAFLD). In this new effort, the researchers studied mouse models (mice engineered to develop NAFLD and liver cancer) to better understand how NAFLD-related cancers develop. They note that such research has become more urgent as NAFLD-related liver cancers have become more prevalent in recent years.
The team started with prior research results showing that one particular type of liver cancer, hepatocellular carcinoma, is particularly prevalent in obese people. Suspecting that it is tied to the SQLE gene, which causes the creation of the enzyme squalene epoxidase in the body, the researchers collected RNA samples from tumor tissue in mouse models and from tissue near the same tumors. They found that SQLE was over-expressed in the tumor tissue as compared to wild mice. They also found that mice that over-expressed the enzyme were more likely to develop liver cancer.
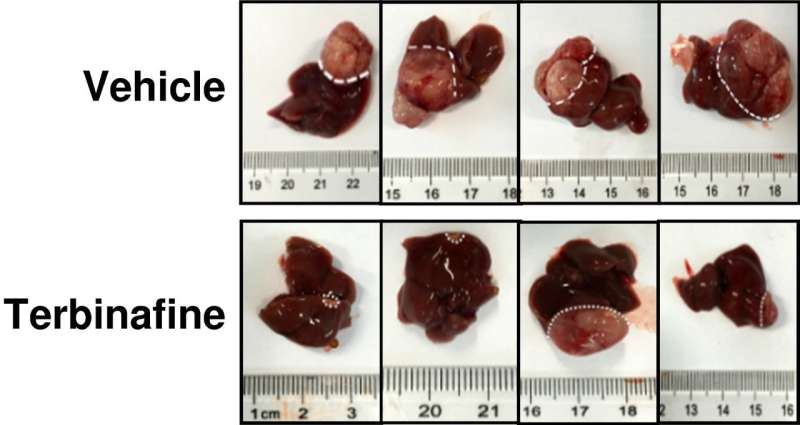
The researchers also found that NAFLD-related tumors could be treated with a compound called terbinafine, which is often used to treat athlete's foot—giving it to the mice, the researchers found, caused reductions in tumor size. Cha, Coates and Brown suggest that the results found by the team in China might lay the groundwork for future work geared towards learning more about the role SQLE plays in liver (and possibly other) cancers and other possible treatments for it.
More information: D. Liu el al., "Squalene epoxidase drives NAFLD-induced hepatocellular carcinoma and is a direct pharmaceutical target," Science Translational Medicine (2018). stm.sciencemag.org/lookup/doi/ … scitranslmed.aap9840
Abstract
Nonalcoholic fatty liver disease (NAFLD)–induced hepatocellular carcinoma (HCC) is an emerging malignancy in the developed world; however, mechanisms that contribute to its formation are largely unknown, and targeted therapy is currently not available. Our RNA sequencing analysis of NAFLD-HCC samples revealed squalene epoxidase (SQLE) as the top outlier metabolic gene overexpressed in NAFLD-HCC patients. Hepatocyte-specific Sqle transgenic expression in mice accelerated the development of high-fat, high-cholesterol diet–induced HCC. SQLE exerts its oncogenic effect via its metabolites, cholesteryl ester and nicotinamide adenine dinucleotide phosphate (NADP+). Increased SQLE expression promotes the biosynthesis of cholesteryl ester, which induces NAFLD-HCC cell growth. SQLE increased the NADP+/NADPH (reduced form of NADP+) ratio, which triggered a cascade of events involving oxidative stress–induced DNA methyltransferase 3A (DNMT3A) expression, DNMT3A-mediated epigenetic silencing of PTEN, and activation of AKT-mTOR (mammalian target of rapamycin). In human NAFLD-HCC and HCC, SQLE is overexpressed and its expression is associated with poor patient outcomes. Terbinafine, a U.S. Food and Drug Administration–approved antifungal drug targeting SQLE, markedly inhibited SQLE-induced NAFLD-HCC cell growth in NAFLD-HCC and HCC cells and attenuated tumor development in xenograft models and in Sqle transgenic mice. Suppression of tumor growth by terbinafine is associated with decreased cholesteryl ester concentrations, restoration of PTEN expression, and inhibition of AKT-mTOR, consistent with blockade of SQLE function. Collectively, we established SQLE as an oncogene in NAFLD-HCC and propose that repurposing SQLE inhibitors may be a promising approach for the prevention and treatment of NAFLD-HCC.
© 2018 Medical Xpress