New paper suggests that tumor cells may develop resistance to potential Ras inhibitors
Researchers have long sought a treatment aimed specifically at the Ras family of genes, the most common oncogenes and those that initiate many of the most lethal tumours. However, the results of a hypothetical treatment may be far less positive than speculated, according to a manuscript published in Genes & Development by the Genomic Instability Group at the Spanish National Cancer Research Centre (CNIO). The study shows that cells are capable of surviving even in the total absence of Ras genes if another gene, Erf, is also lost.
Discovered in 1982 by Mariano Barbacid's group, among others, alterations in Ras genes were the first mutation described in cancer. This was a paradigm-shifting discovery, revealing for the first time that tumours are initiated by mutations in genes, thereby raising hope that inhibitors for these mutated genes could cure cancer. "It is the basis of personalised medicine," explains Óscar Fernández-Capetillo, leader of this work.
Mutations in Ras genes lead to the most lethal tumours, including lung, pancreas and colon cancer. Therefore, developing a RAS pharmacological inhibitor has frequently been described as the search for the Holy Grail in the battle against cancer, in which billions have been invested. However, achieving an inhibitor for RAS proteins is complicated due mainly to their three-dimensional structure, similar to a sphere, which makes it difficult to generate pharmaceuticals that inhibit their activity. As an alternative to treating these tumours, pharmaceuticals have been developed that attack the other members of the Ras route, such as inhibitors of MEK, RAF and EGFR.
"Personalised medicine, in spite of its success stories, has an Achilles heel in the fact that tumours have dozens or even hundreds of mutations, so while treatments generally work for a limited time, tumours invariably end up developing resistance due to another mutation," says Fernández-Capetillo.
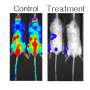
Although therapies against the RAS route constitute an important part of current antitumoral strategies, the search for RAS inhibitors continues "despite the fact that it is not clear if tumours are not going to be capable of developing resistance to these treatments," says Sergio Ruiz, co-leader of the study. "In our work, we show that it is even possible to develop teratomas (a type of germinal tumour) lacking all RAS genes, if the tumour also lacks ERF expression."
ERF loss rescues the effects of RAS deficiency
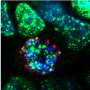
The main role of RAS proteins consists of translating external growth signals (nutrients, growth factors, etc.) into proliferating responses within the cell. When RAS proteins are eliminated in mouse stem cells, these remain in a sort of suspended state—they do not grow, they do not differentiate into other cell types and are not capable of forming tumours.
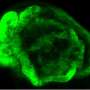
Cristina Mayor-Ruiz, first author of the study, initially observed that certain tumour cells were capable of growing even in the absence of serum, if the gene Erf was also eliminated. Fernández-Capetillo explains "For me, this discovery was the origin of the project as it made us speculate that if cells can grow with hardly any nutrients upon eliminating ERF, this could even allow the growth of RAS-free cells." This hypothesis turned out to be correct—eliminating ERF allows mouse embryonic stem cells to grow, differentiate and even generate tumours in total absence of RAS genes.
The study also explains the mechanism by which ERF restricts the action of RAS proteins. In the absence of RAS, ERF is recruited to the regulating areas ("enhancers") of multiple genes, modulating their function, which ultimately limits cell growth. "ERF is a kind of brake that limits the consequences of RAS activation," indicates Cristina Mayor-Ruiz.
"The message is not good, but its knowledge is important for cancer research and personalised medicine," says Fernández-Capetillo. "Although a perfect inhibitor of RAS has been achieved, tumours may be capable of resistance to the treatment, accumulating mutations in genes like ERF." In fact, recent studies have found ERF mutations in cancer patients, indicating that this situation may, indeed, exist in the clinic. Therefore, Fernández-Capetillo's group is now exploring whether mutations in ERF can account for the resistance to personalised therapies against inhibitors of the RAS route.
More information: Cristina Mayor-Ruiz et al, ERF deletion rescues RAS deficiency in mouse embryonic stem cells, Genes & Development (2018). DOI: 10.1101/gad.310086.117