Study identifies new potential treatment option for triple negative breast cancer
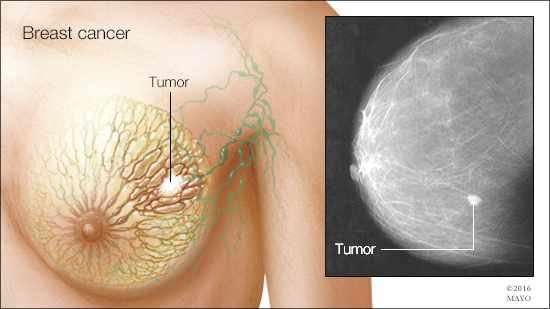
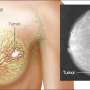
In a recent study published in the Journal of Clinical Investigation, Mayo Clinic researchers identified that an FDA drug approved for myelodysplastic syndrome may be useful to treat triple-negative breast cancer, which is one of the most aggressive and lethal types of breast cancer.
In this study, Mayo investigators identified that the drug 5-aza-2'-deoxycytidine (decitabine) which is FDA approved for the treatment of certain hematological (blood) cancers, could significantly inhibit the growth of triple-negative breast cancers, and importantly this effect was also seen in tumors resistant to chemotherapy. This response was dependent on the presence of certain critical proteins called DNA methyl transferase proteins that are present in only a subset of triple negative breast cancers. This provides a way to identify which patients could benefit from this therapy.
"There is a great need to identify additional treatment options for triple-negative breast cancer, which is one of the most difficult to treat subtypes of breast cancer," says Mayo researcher Liewei Wang, M.D., Ph.D. "The study is a demonstration that we can take advantage of many existing FDA approved drugs to expand their usage by better understanding the mechanisms of how they work and applying them to other cancers."
This study was part of the ongoing work from the Breast Cancer Genome-Guided Therapy (BEAUTY) study, co-led by Matthew Goetz, M.D., a Mayo medical oncologist and Judy Boughey, M.D., a Mayo breast surgeon. The BEAUTY study generated patient derived xenografts (immortalizing breast tumor cells) from patients with breast cancer treated with chemotherapy.
"Patients whose tumor does not respond well to chemotherapy are known to be at significantly increased risk of recurrent breast cancer and death," says Dr. Boughey. "Therefore our focus is to identify new treatment options for these patients."
In the living xenografts from BEAUTY, Mayo investigators found that when DNA methyl transferase proteins were present, decitabine showed an effect in triple-negative breast cancer at a low therapeutic dose. The low doses would result in less toxicity and might allow the drug to be used for a longer time, all of which might help to achieve greater therapeutic efficacy.
According to Dr. Goetz, plans are underway to prospectively study the impact of decitabine in a prospective clinical trial, called BEAUTY2, which is focused on women with triple negative breast cancer that is resistant to chemotherapy.