Cross-species prion adaptation depends on prion replication environment
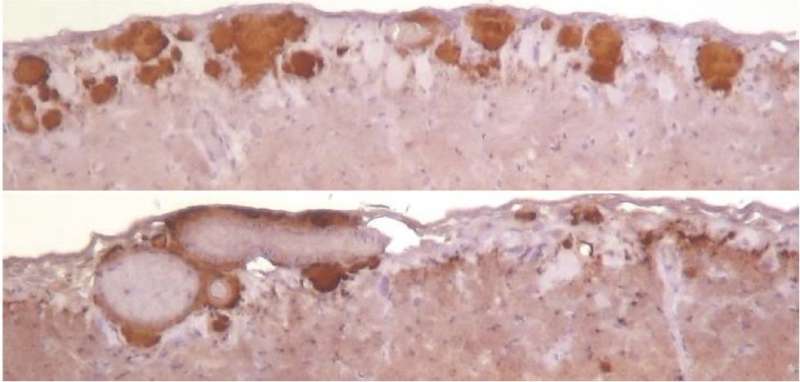
A hamster prion that replicated under conditions of low RNA levels in mouse brain material resulted in altered disease features when readapted and transmitted back to hamsters, according to new research presented in PLOS Pathogens by Elizaveta Katorcha of the University of Maryland School of Medicine, Baltimore, and colleagues.
Prions are abnormal proteins that can induce normal versions of the protein to mis-fold, leading to neurodegenerative disease. Prion diseases in humans, such as bovine spongiform encephalopathy ("mad cow disease") can arise via cross-species transmission of prions from other mammals. Whether a prion will jump and adapt to a new species depends on the specific amino acid sequences of the proteins involved. However, previous research suggests that other factors may be in play.
The author's lab had previously found that replication of prions in an environment with low levels of RNA, without changing their amino acid sequence, results in changes to their biological activity. In the new study, they investigated how low-RNA replication environments affect cross-species transmission.
The researchers propagated a hamster prion known as 263K under conditions of low RNA levels in mouse brain material. Then, they transferred it to hamster brain material and propagated it under normal RNA conditions. When the adapted strain was subsequently transferred to live hamsters, it resulted in disease that lasted longer and had different neuropathological features than normal 263K disease.
Disease caused by 263K propagated under normal RNA conditions in mouse and then hamster brain material appeared similar to normal disease. Meanwhile, 263K propagated in RNA-depleted mouse and then RNA-depleted hamster brain material failed to cause disease in hamsters.
These results suggest that prion adaptation during cross-species transmission depends, in part, on surrounding RNA levels during replication. While RNA is not incorporated into the structure of prions, it could be that its unique structure and sites of negative electrical charge influence prion replication. The new findings could help improve understanding of cross-species prion transmission and the factors that control prion adaptation and disease in mammals.
More information: Katorcha E, Gonzalez-Montalban N, Makarava N, Kovacs GG, Baskakov IV (2018) Prion replication environment defines the fate of prion strain adaptation. PLoS Pathog 14(6): e1007093. doi.org/10.1371/journal.ppat.1007093