Alzheimer's disease and type 2 diabetes—hope for inhibitors against amyloid plaques
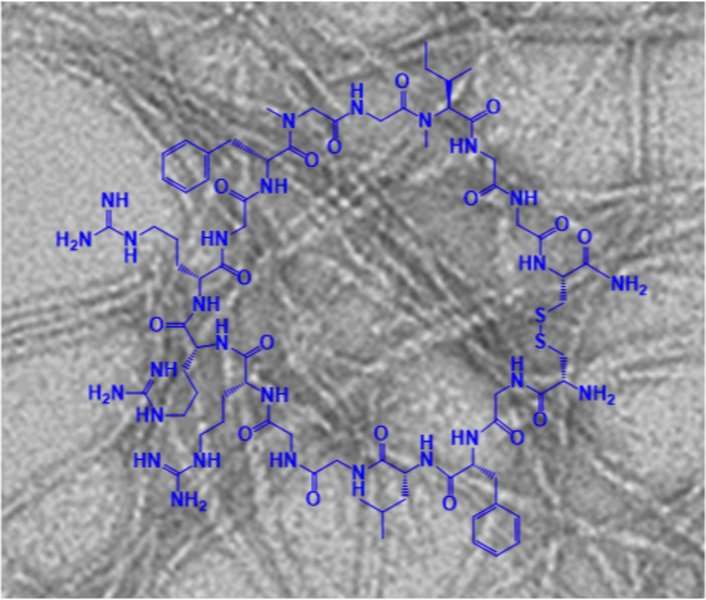
Effective therapeutics to counteract the formation of amyloid plaques in Alzheimer's disease and type 2 diabetes are not yet available. Scientists at the Technical University of Munich (TUM) have now come a little bit closer to a solution: They have described a new class of designed macrocyclic peptides that are highly potent inhibitors of amyloid formation.
Amyloid plaques, which are protein deposits, play a crucial role in the development of Alzheimer's disease and type 2 diabetes. Several teams of scientists around the world are working on finding ways to prevent amyloid plaque formation in the human brain.
The research team of Aphrodite Kapurniotu, Professor for Peptide Biochemistry (TUM) has been working on an idea for some time now in collaboration with the teams of Professor Martin Zacharias (TUM), Professor Gerhard Rammes (TUM Rechts der Isar Hospital) and Professor Jürgen Bernhagen (Institute for Stroke and Dementia Research (ISD) at Ludwig Maximilian University (LMU)). The researchers have presented macrocyclic peptides (MCIPs) as potent inhibitors of amyloid formation. They reported their results in the journal Angewandte Chemie.
New class of amyloid inhibitors
In its new study, the team presents macrocyclic peptides, developed as a new class of amyloid inhibitors. "We have discovered an MCIP that is stable in human blood plasma and can also overcome the human blood-brain barrier in an in vitro cell culture model," explains Professor Kapurniotu. She adds, "So far we were only able to demonstrate these properties in the test tube—thus, further research is necessary. But these are two highly desirable properties for inhibitors of Alzheimer's amyloid."
TUM has already applied for a patent for the newly developed macrocyclic peptides. "They could be a good alternative to the currently pursued antibody-based approaches as therapeutics against Alzheimer's amyloid plaque formation because they are easy to produce, have promising properties and, due to their peptidic nature, they will be significantly cheaper than antibodies," says Professor Kapurniotu.
"Therefore, further investigations are now planned to verify whether the MCIPs are also effective in in vivo models. Furthermore, the MCIPs could also be suitable as templates for the development of small molecule peptidomimetics (molecules mimicking peptide chains), which might also find application as anti-amyloid drugs in Alzheimer's and type 2 diabetes.
More information: Anna Spanopoulou et al, Designed Macrocyclic Peptides as Nanomolar Amyloid Inhibitors Based on Minimal Recognition Elements, Angewandte Chemie International Edition (2018). DOI: 10.1002/anie.201802979