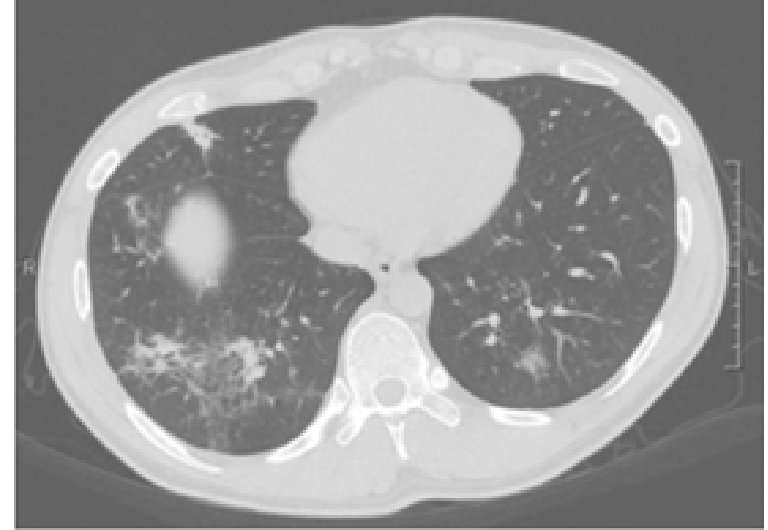
CT scans show that the excessive inflammation and infection caused by the mutation in the IKBKB gene results in the thickening of bronchi in the lungs. Credit: Cardinez et al., 2018
Researchers from Australia and Japan have discovered a new human immunodeficiency syndrome in two patients on separate continents. The study, which will be published October 18 in the Journal of Experimental Medicine, reveals that a mutation in a gene called IKBKB disrupts the immune system, leading to excessive inflammation and the loss of both T and B white blood cells.
Genetic mutations can cause a wide variety of severe, inheritable immune deficiencies that begin at birth or in early childhood. But the causes of other, more sporadic, immune diseases, including milder immunodeficiencies that can develop later in life, remain poorly understood. A team of researchers led by Matthew C. Cook from Australian National University in Canberra and Hirokazu Kanegane from Tokyo Medical and Dental University analyzed a group of immunodeficient patients in Australia and Japan and identified two unrelated patients—one in each country—carrying an identical mutation in a gene called IKBKB.
Both patients seemed to have developed the mutation "de novo," rather than inheriting it from their parents. Their symptoms were slightly different, but they were both susceptible to recurrent infections and showed signs of excessive inflammation. Both patients had reduced numbers of antibody-producing B cells and two types of effector T cells that help the body fight off infections. However, the T cells that remained appeared to be more prone to activation.
The IKBKB gene encodes an enzyme called IKK2 that controls the development and activation of white blood cells by regulating the NF-kB cell signaling pathway. Mutations that inhibit IKK2 and NF-kB signaling are known to cause severe early onset immune deficiencies. But the mutation found in the two Australian and Japanese patients seems to result in a hyperactive form of IKK2 that increases NF-kB cell signaling.
To confirm that this mutation was responsible for the patients' symptoms, the researchers used CRISPR/Cas9 gene editing to create the same mutation in mice. These animals also showed an increase in IKK2 and NF-kB cell signaling and developed similar defects in their T and B cells.
"Our results show that a de novo mutation in human IKBKB increases the function of IKK2 and results in a combined immune deficiency syndrome affecting both T and B cells," Kanegane says.
"We were able to confirm this by engineering an equivalent mutation in mice," says Cook. "In future, this strategy should prove important for investigating therapeutic interventions for personalized therapy."
More information: Journal of Experimental Medicine (2018). DOI: 10.1084/jem.20180639
Journal information: Journal of Experimental Medicine
Provided by Rockefeller University Press