Monthly buprenorphine injections effective for opioid use disorders
A newly published study suggests the long-acting buprenorphine formulation BUP-XR is more effective than placebo in helping patients struggling with addiction to opioids. BUP-XR is a monthly extended-release injection.
Daily dose buprenorphine was approved by the U.S. Food and Drug Administration in 2002 and has proven to be effective in the treatment of opioid use disorders (OUD). However, daily doses require patients to recommit to taking medication every day and can result in cravings near the end of the 24-hour cycle. Although the National Institute on Drug Abuse (NIDA) did not fund this current study, it did support the development of buprenorphine as a treatment for OUD. NIDA's support also contributed to the development and approval of another long-acting buprenorphine formulation—a six-month subcutaneous implant for some patients who have stabilized on daily dosing (Probuphine).
In November 2017, the FDA approved the use of an extended release buprenorphine (BUP-XR) under the brand name Sublocade as a monthly long-acting injection, based on positive results in a Phase III human subjects study. While data that underlie FDA approval decisions are proprietary and not always available to the public, the study has now been published in The Lancet to make it available to the wider scientific community.
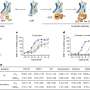
In the randomized, double-blind study, 200 participants with a mean duration of opioid use of 11-12 years were divided into three groups, two with different monthly doses of BUP-XR and one with placebo. Both BUP-XR regimens led to substantial proportions of participants achieving abstinence from opioids, relief of withdrawal symptoms, and control of opioid craving without the need for daily medication adherence or supplemental buprenorphine. Retention was nearly twice as high with BUP-XR compared with placebo.
More information: For more information on medication-assisted treatment for opioid use disorder, go to www.drugabuse.gov/publications … nts-opioid-addiction
Barbara R Haight et al. Efficacy and safety of a monthly buprenorphine depot injection for opioid use disorder: a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial, The Lancet (2019). DOI: 10.1016/S0140-6736(18)32259-1