A billion years of evolution suggests a new approach for Alzheimer's disease treatment

New research by academics at Royal Holloway, University of London, urges scientists that are looking to finding a cure for Alzheimer's disease to focus on the role of proteins in the brain that cause Alzheimer's disease. This change in focus could enable the development of new treatments.
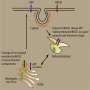
It has long been recognised that genetically inherited (familial) Alzheimer's disease is caused by mutations in a group of proteins within the human brain known as the gamma secretase complex. The role of these proteins was first identified when mutations in the proteins caused the formation of plaque deposits found in the brains of Alzheimer's patients, leading to the death of nerve cells in the brain. Scientists have spent many decades analysing this effect without identifying effective treatments.
Alternatively, some researchers have suggested that the gamma secretase complex also control a process called autophagy. Autophagy is a process where proteins are broken down into amino acids and reused within the cell, and when autophagy is blocked by mutations in the gamma secretase complex in Alzheimer's disease, cells in the brain die.
New research led by Professor Robin SB Williams and Dr. Devdutt Sharma from the Centre for Biomedical Sciences at Royal Holloway, has found that the gamma secretase complex also controls autophagy in a single-celled organism amoeba, known as Dictyostelium, which shared a common ancestor with humans around a billion years ago. The conservation of this role in autophagy for all these years illustrates its critical importance in cell health. This therefore suggests that researchers should look at this process to develop new treatments for Alzheimer's disease.
During this research, Professor Williams and his group used the amoeba, Dictyostelium, as a model to investigate the role of the gamma secretase complex. The group was able to delete individual proteins from the complex, such as the presenilin protein, and monitor changes in cell function. They found that when proteins from the gamma secretase complex were deleted, the process of autophagy was blocked, and so cells were unable to recycle proteins.
Professor Williams, said: "These findings suggest that the role of the gamma secretase complex is conserved across a huge evolutionary distance, indicating a fundamentally important role in maintaining healthy cells. From this, we propose that researchers looking to find new approaches to treat patients with Alzheimer's disease should focus on restoring normal levels of autophagy in neurons of Alzheimer's disease patients to block the death of brain cells in these patients.
"Up until now, there has been very little progress in finding a cure for Alzheimer's disease. However, if scientists begin to take a different approach to investigating this illness, we may be able to find a way to block the disease. Whilst current medication slows the development of some symptoms, these are not long term solutions, but simply delay the disease progression.
"We are hopeful that, using our innovate approach to this research, we will help to refocus researchers in this area, to successfully develop new treatments for patients with Alzheimer's disease."
More information: Devdutt Sharma et al. Gamma secretase orthologs are required for lysosomal activity and autophagic degradation in Dictyostelium discoideum, independent of PSEN (presenilin) proteolytic function, Autophagy (2019). DOI: 10.1080/15548627.2019.1586245