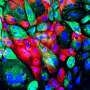
RNA transcribed from genes that seem not to code for anything may play an important role in regulating cancer, a new study suggests.
A number of these noncoding RNA fragments lie next to known cancer genes, the study found. Understanding how they interact with those cancer genes could open new avenues to understanding cancer's behavior and treating it.
Led by Kannanganattu Prasanth, a University of Illinois cell and developmental biology professor, the researchers found that a certain breast cancer tumor suppressor gene needs the RNA transcribed from its opposite DNA strand to function in healthy cells. They published their work in the journal PLOS Genetics.
When a gene is expressed, the two strands of DNA unzip and one of the strands is transcribed into RNA. The RNA then travels out of the nucleus to be translated into protein. However, only a small fraction of the RNA strands are translated into proteins. The rest, called noncoding RNAs, were once thought to be "junk" but now are known to play several roles in the cell, such as regulating the expression of genes that code for proteins.
"We know a lot of the mutations in breast cancer cells happen to be on noncoding regions of the genome," Prasanth said. "If these mutations are contributing to cancer, but are not coding for a protein, what are they doing?"
First, the researchers analyzed RNA expression in cells of a type of breast cancer known as triple-negative breast cancer, and in healthy breast cells. They found significant differences in how the cells expressed more than 1,800 long noncoding RNAs. About 500 of those were transcribed from locations on the opposite DNA strand from known protein-coding genes. Such RNAs are called "antisense," since they are transcribed from the strand that lies opposite the coding or "sense" DNA fragment.
"Several antisense RNAs showed correlation with their neighbor protein-coding genes. Among these protein-coding genes, there are several cancer-related genes with known functions in tumor progression," said Mahdieh Jadaliha, the first author of the paper and a recent graduate from Prasanth's group. "The correlation between these cancer genes and their antisense RNAs could be a clue that the RNAs could potentially function in cancer progression through regulating the expression of these cancer-associated genes."
The researchers focused on the antisense RNA to a well-studied tumor-suppressor gene called PDCD4, which is somehow deactivated in triple-negative breast cancer cells. They wondered if the reduced levels of PDCD4 antisense RNA they saw in breast cancer cells could contribute to the PDCD4 gene deactivation.
In studying the molecular mechanisms of both PDCD4 RNA and its antisense counterpart, they found that both were crucial for the PDCD4 gene to function in normal breast cells.
"We noticed that the PDCD4 antisense RNA stabilizes the protein-coding PDCD4 RNA by binding to it and protecting it from being degraded in the cell," Prasanth said.
Without the antisense RNA, the PDCD4 RNA was degraded before it could be translated into protein. The gene could not perform its role of stopping the cancer cell from proliferating.
When the PDCD4 antisense was restored in cancer cell cultures, the cells' growth was inhibited and they were less able to migrate and spread, the team found.
"This suggests that in patients where PDCD4 tumor suppressor activity is low, we potentially could increase its activity by activating the PDCD4 antisense RNA. In theory, we should be able to enhance the expression of the tumor suppressor in cancer cells," Prasanth said.
The researchers say the example of PDCD4 shows that mutations in genes that seem to be noncoding could help fill in gaps in understanding cancer's mechanisms, as well as how gene expression is regulated in the cell. Prasanth's group members plan to study more of the antisense RNAs they identified as possibly playing a role in triple-negative breast cancer. They also plan to continue to study PDCD4 and its antisense RNA in animal models.
"This is one part of a big puzzle," Prasanth said. "We are seeing 500 different antisense RNAs being deregulated in cancer. Thirty or 40 of them seem to be antisense to well-established tumor suppressor and oncogenes. But what about the other 460? By understanding their function, we may be able to identify new oncogenes or novel mechanisms for tumor suppression. It's an uncharted territory, which is challenging and exciting at the same time."
More information: Mahdieh Jadaliha et al. A natural antisense lncRNA controls breast cancer progression by promoting tumor suppressor gene mRNA stability, PLOS Genetics (2018). DOI: 10.1371/journal.pgen.1007802
Journal information: PLoS Genetics
Provided by University of Illinois at Urbana-Champaign