Clinical trial explores opening blood-brain barrier in fight against Alzheimer's
A new clinical trial at The Ohio State University Wexner Medical Center and two other sites is testing an innovative procedure that may provide hope in the fight against Alzheimer's disease.
The non-invasive procedure uses low-intensity focused ultrasound to open the blood-brain barrier, a protective layer that protects the brain from infections or pathogens in the blood. However, this barrier also makes it nearly impossible to deliver therapeutics to the brain to treat neurodegenerative diseases such as Alzheimer's.
"While it's protective and beneficial for day-to-day brain function, when we think about therapeutics, the blood-brain barrier poses a significant challenge," said Dr. Vibhor Krishna, a neurosurgeon at Ohio State Wexner Medical Center. "The focused ultrasound procedure allows us to non-invasively access the brain tissue so treatment can be administered straight to the site of pathology."
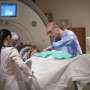
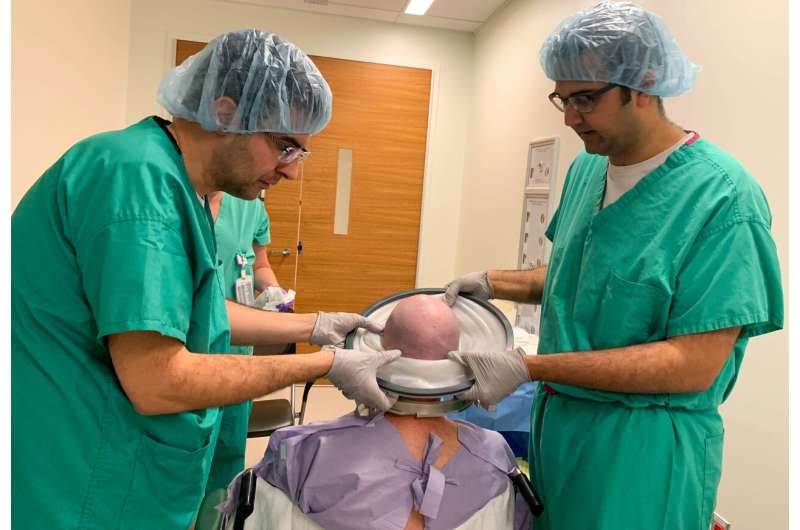
During the surgery in an intraoperative MRI-surgical suite, patient David Shorr of Bexley, Ohio, was awake and alert, giving real-time feedback to the treatment team of neurosurgeon, neurologist, neuropsychologist, researchers and nurses.
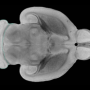
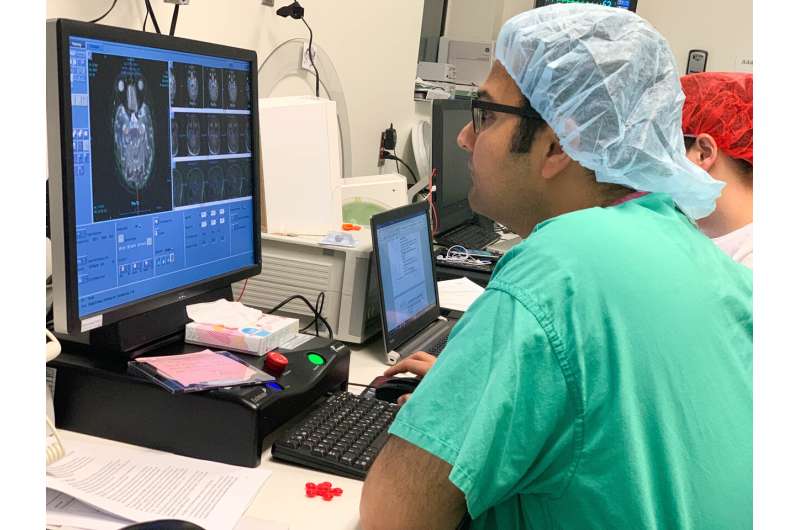
MRI-guided imaging allows doctors to target a specific area of the brain where there is a buildup of toxic proteins called amyloid. A head frame holds the patient's head still during the treatment. The ultrasound transducer—essentially a helmet housing the ultrasound beams—is attached to the head frame.
Surrounding the patient's head is a water bath where cold, degassed water is circulated. This setup helps transmission of 1,000 focused ultrasound beams from the machine through intact skull focused at the exact brain tissue that is being targeted.
During the procedure, the patient's bloodstream is infused with microbubbles. The focused ultrasound waves are delivered through the helmet-like device, which causes microbubbles in the blood to oscillate and open the blood-brain barrier.
"In this research study, we are not delivering any medications. Our hypothesis is that, by opening the blood brain barrier, a patient's own immune defense may clear some of those harmful amyloids," said Krishna. "If we determine this to be safe, in the next steps we would want to understand the effectiveness and the impact of opening the blood-brain barrier in improving cognitive decline."
The procedure is performed three times at two-week intervals to allow for as much amyloid clearance as possible. In the future, this method of opening the blood-brain barrier may also be applicable in developing new treatments for brain tumors and epilepsy.
"With 5.8 million Americans living with Alzheimer's, there's a critical need to develop novel therapies to treat this devastating disease. With this innovative clinical trial, Ohio State researchers are pioneering potential new treatments," said Dr. K. Craig Kent, dean of the College of Medicine.
The research team at Ohio State's Center for Neuromodulation will monitor the patients closely, using neurological exams and neuro-psychological exams to assess language, memory and executive functioning at various intervals for one year following the surgery.
The clinical trial, sponsored by Insightec, will enroll up to 10 patients at Ohio State Wexner Medical Center, Weill Cornell Medicine and West Virginia University Rockefeller Neuroscience Center.
"We're hopeful it can help him, but we also know maybe it will help somebody else," said Shorr's wife, Kim Shorr.