The mechanism behind the pluripotency-differentiation transition
Induced pluripotent stem cells can turn into any type of cell in the body or remain in their original form. In the current edition of Molecular Cell, scientists from the Helmholtz Zentrum München describe how cells decide which of these two directions to take. During their research, they identified a protein and a ribonucleic acid (RNA) that play a highly significant role in this process. Their discovery also allows a better understanding of amyotrophic lateral sclerosis (ALS), a progressive neurological disease that affects motor neurons.
Thanks to their ability to transform into any type of cell in the body, induced pluripotent stem cells (iPS cells) could make a key contribution to regenerative medicine. In order to generate artificial beta cells for the treatment of type 1 diabetes, for example, it is essential to understand the mechanisms that underlie cell differentiation. Together with his team, Dr. Micha Drukker from the Institute of Stem Cell Research (ISF) at the Helmholtz Zentrum München, has now shown how such processes are controlled at the molecular level. It all began with a structure in a cell nucleus that was visualized with the aid of fluorescence microscopy.
Two key factors in the cell nucleus
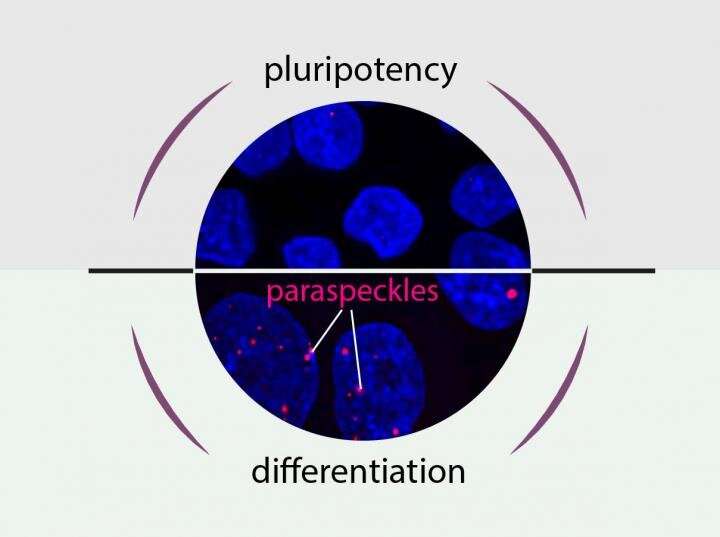
"We noticed that nuclear domains named paraspeckles do not occur in iPS cells, but are quickly formed during the differentiation process, irrespective of the cell type that we created," says Dr. Miha Modic, who was a member of Dr. Drukker's working group. Drukker and Modic hypothesized that this phenomenon was linked to the ability of stem cells to differentiate into body cells. Along with Prof. Ule Jernej from University College London and Markus Grosch, a Ph.D. student in Dr. Drukker's group, the researchers discovered key molecules in the cell nucleus that orchestrate the appearance of paraspeckles and how they regulate differentiation.
"Two factors play key roles in the decision as to whether cells differentiate or remain pluripotent," says Drukker. "We identified NEAT1, a ribonucleic acid (RNA), and TDP-43, a protein that binds to RNA." NEAT1 exists in two forms. The short form is stabilized by TDP-43, and in this case, no paraspeckles develop. The cell remains pluripotent, and is not altered. Conversely, a decrease in TDP-43 creates the long form of NEAT1. Paraspeckles are formed, and an iPS cell begins to differentiate. Modic adds: "This control system may be general for stem cells to make a choice when to differentiate."
Modic says, "Dr. Silvia Schirge and Prof. Heiko Lickert from the Institute of Diabetes and Regeneration Research (IDR) helped us to show that paraspeckles are also crucial for efficient differentiation during murine embryonic development. Together, their study provides a breakthrough in understanding processes of differentiation and development.
Link to diseases
In Drukker's view, these findings will do more than just provide a contribution to basic research. "Paraspeckles are linked to many diseases, but up until now they have rarely been examined in the context of developmental and stem cell biology," he explains. In the case of amyotrophic lateral sclerosis (ALS), the role of TDP-43—as well as the appearance of paraspeckles—is particularly evident. In motor neurons, the cells that operate our muscles and are affected by ALS, TDP-43 is oddly regulated and forms toxic aggregates; and there is an increased occurrence of the long form of NEAT1, and more paraspeckles can be detected. These mechanisms are regarded as an early sign of ALS—even before patients present with clinically relevant symptoms. In the next stage, Drukker and his team of scientists hope to examine other cell types for paraspeckles, RNAs and their interactions. By then it will also be apparent whether the newly discovered molecules will provide suitable targets for pharmacotherapies.
More information: Miha Modic et al, Cross-Regulation between TDP-43 and Paraspeckles Promotes Pluripotency-Differentiation Transition, Molecular Cell (2019). DOI: 10.1016/j.molcel.2019.03.041