Topical cream found as less-toxic therapy to treat cutaneous leishmaniasis
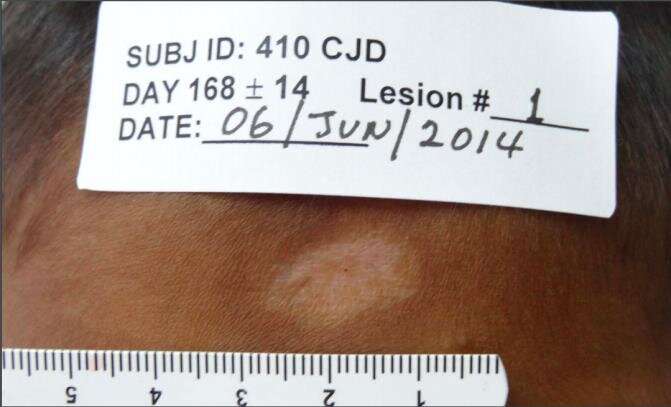
Paromomysin-based topical treatments were shown to be effective in curing cutaneous leishmaniasis (CL), according to a randomized, double blind study conducted in Panama and published with PLOS Neglected Tropical Diseases.
Leishmaniasis, transmitted by a female phlebotomine sand fly bite, is endemic in 98 countries and has approximately 0.7 to 1.2 million CL cases each year. Infection with CL causes open and chronic lesions on the skin and joints, as well as debilitating pain. Many patients who have CL in Panama can receive pentavalent antimony, (compounds that are injected intravenously or intramuscularly) however this treatment can be toxic and can prevent the patient from receiving full care. In addition, many patients live in areas where this laboratory treatment is inaccessible.
Néstor Sosa, Juan Miguel Pascale and colleagues, ran a study to see if a topical cream could cure this disease in a less toxic and more accessible way. The researchers tested two creams, one with only paromomysin, an antimicrobial, and one with a combination of paromomysin and gentamicin (gentamicin is an antibiotic that stops the further growth of bacteria). The addition of gentamicin did not change the cure rate for the 399 patients treated over 20 days. Results showed that there was a high cure rate of close to 80%. Children under 12, and teens from 12-17, had an especially high cure rate of 84% and 82% respectively. However, not all patients were cured: 87 patients experienced either disease persistence, disease worsening or disease relapse, due to a lack of the disease's response to the antibiotics by Day 100 of the test. 16 other patients were clinical failures due to withdrawal of consent or lost follow-up.
"This trial demonstrates that topical therapy with a paromomycin-based cream offers a potential alternative to the current standard of care for the treatment of CL in Panama," the authors note. "A topical treatment for uncomplicated CL is also of great interest in the United States, where current treatment options all have suboptimal risk/benefit profiles for this disease."
More information: Sosa N, Pascale JM, Jiménez AI, Norwood JA, Kreishman-Detrick M, et al. (2019) Topical paromomycin for New World cutaneous leishmaniasis. PLOS Neglected Tropical Diseases 13(5): e0007253. DOI: 10.1371/journal.pntd.0007253














