Is multiple sclerosis linked to childhood viral infections?
Although the exact causes of multiple sclerosis still remain unknown, it is assumed that the disease is triggered by a combination of genetic and environmental risk factors. But which? In a mouse model of the disease, researchers at the University of Geneva (UNIGE) and the Geneva University Hospitals (HUG), Switzerland, studied the potential link between transient cerebral viral infections in early childhood and the development of this cerebral autoimmune disease later in life. Indeed, the brain area affected by viral infection during childhood undergoes a change that can call, a long time later, on the immune system to turn against itself at this precise location, triggering autoimmune lesions. These results, which are published in the journal Science Translational Medicine, provide a first step in answering one of the possible environmental causes of this serious disease.
Multiple sclerosis affects one in 1,000 people in Switzerland, two-thirds of whom are women. It is the most common auto-immune disease affecting the brain. Up to date, there is still neither a cure available, nor a clear understanding of the factors that trigger this disease at around 30 years of age. "We asked ourselves whether brain viral infections that could be contracted in early childhood were among the possible causes," says Doron Merkler, a professor in the Department of Pathology and Immunology in UNIGE's Faculty of Medicine and senior consultant in the Clinical Pathology Service of the HUG. Such transient brain infections can be controlled quickly by the immune system, without the affected individual even noticing any symptoms. "But these transient infections may, under certain circumstances, leave a local footprint, an inflammatory signature, in the brain," continues the researcher.
The childhood: a pivotal moment influencing disease risk
The scientists induced a transient viral infection in a group of adult mice and in a group of mice at a very young age in order to test this hypothesis. Karin Steinbach, a researcher in the same department, explains: "In both cases, the mice showed no signs of the disease and eliminated the infection within a week with a similar anti-viral immune response."
The scientists then allowed the two groups of mice to grow older before they were transferred with self-reactive cells, which can target the normal brain structure and are also thought to contribute to the illness of patients with multiple sclerosis. "These self-reactive cells are present in most of us, but do not necessarily induce a disease, since they are controlled by different regulatory mechanisms and usually don't have access to the brain," explains Karin Steinbach.
Indeed, in the group of mice infected with the virus in adulthood, the transferred self-reactive cells did not gain access to the brain and no brain lesions were observed. However, in those mice that had been infected at a very young age, the self-reactive cells gained access to the brain in adulthood, and migrated to the precise location where the infection had previously occurred. As a result, self-reactive cells started to attack the brain structure in these areas, leading to the development of brain lesions. Why was there such a difference depending on the age at which the mice suffered a prior viral infection?
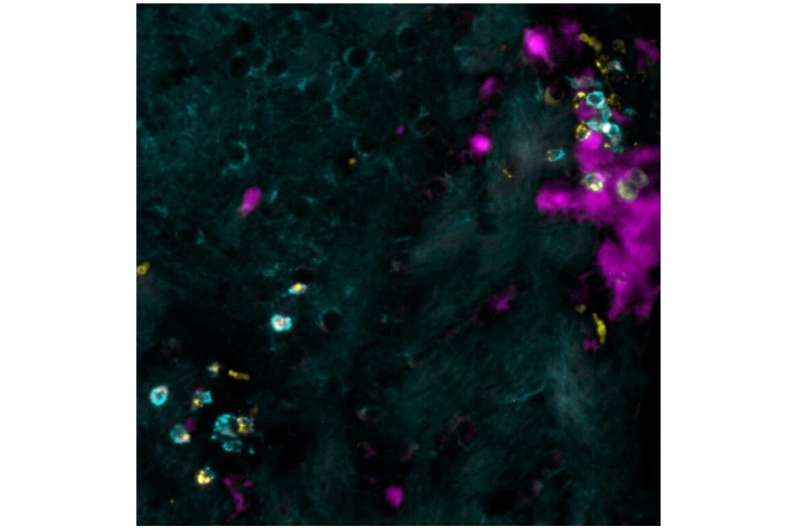
An accumulation of T cells gives the signal
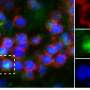
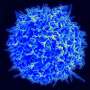
During their analysis of the brains in the cohort of mice that had overcome the viral infection at a very young age, the researchers observed an accumulation of a sub-type of immune cells: so-called "brain-resident memory T cells." "Under normal circumstances, these cells are distributed throughout the brain, ready to protect it in case of a viral attack. But here, the cells accumulate in surplus at the exact spot of the infantile infection in the brain," says Professor Merkler. The researchers subsequently found that these cells produced a molecule that specifically attracts the self-reactive cells, allowing them to access the brain and to cause auto-immune brain lesions. "In order to verify this observation," continues the professor, "we blocked the receptor that transmits the signal to the self-reactive cells. Indeed, the mice were then protected from developing brain lesions!"
A similar phenomenon also occurs in humans
"We then looked to see if we could find a similar accumulation of brain-resident memory T cells that produce this molecule in people with multiple sclerosis, and indeed we did," says Karin Steinbach. By analogy, the scientists suggest self-reactive T cells in humans could gain access to the brain by a similar mechanism as observed in mice, something that requires future studies to elaborate on.
"We are continuing our research in this direction. We particularly want to understand why brain-resident memory T cells accumulate in these discrete spots in a child's brain following infection but not in adulthood," concludes Karin Steinbach. In the future, the knowledge gained from these studies may help us understand better the possible causes of multiple sclerosis.
More information: K. Steinbach el al., "Brain-resident memory T cells generated early in life predispose to autoimmune disease in mice," Science Translational Medicine (2019). stm.sciencemag.org/lookup/doi/ … scitranslmed.aav5519