Researchers investigate cells' 'self-eating' as key to preventing, treating obesity, type 2 diabetes
What if we could treat obesity or type 2 diabetes by changing the way our cells take out the trash?
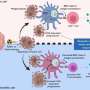
Just as a household can't function well if it's filled with garbage, a cell becomes unhealthy if it can't scrap what it no longer needs. So the cell breaks down these components and uses them as energy, hence the scientific term for the process—autophagy—which comes from the Greek for "self-eating."
A research lab at the University of Florida is investigating how autophagy plays a role in metabolic diseases such as obesity and type 2 diabetes.
"Autophagy is the cell's way of doing quality control. Not enough autophagy, and the cell is not doing well in renewing itself. On the other hand, if you have too much autophagy, it may break the balance (or homeostasis) of cellular components and lead to cell death. So, a finely tuned autophagy is critical for the body to be healthy," Zhiyong Cheng, an assistant professor of human nutrition in the UF Institute of Food and Agricultural Sciences.
Too much or too little autophagy is linked to metabolic diseases, such as obesity, type 2 diabetes, and diabetic complications, Cheng said. "If we can find out what leads to abnormal autophagy, that may reveal new therapeutic options for these diseases. This is the long-standing research interest of my lab," Cheng said.
Autophagy research attracted widespread attention in 2016 when cell biologist Yoshinori Ohsumi won the Nobel Prize in Medicine for his research on autophagy in yeast.
But even before Ohsumi won the Nobel Prize, autophagy research has been part of a growing interest in harnessing the body's own biology to prevent and treat diseases.
Cheng has just published an article reviewing current research on a group of proteins that regulate autophagy. These proteins are called FoxO proteins, and researchers like Cheng see them as a potential key to preventing some of the negative health effects associated with obesity and type 2 diabetes.
"Our research has identified FoxO1, one of the FoxO family, as a regulator of fat tissue formation through autophagy. When we block FoxO1, that prevents the creation of white, energy storing fat, and increases the creation of brown, energy-burning fat," Cheng said. "Because obesity arises from excessive formation and expansion of white fat, targeting FoxO1 may prevent obesity from developing."
The review article examined three major approaches in the current investigation of the FoxO-autophagy relationship.
One approach is to look at how FoxO interacts with DNA. This interaction is called transcription: FoxO binds to DNA so the cell can initiate autophagy. Another approach examines how FoxO functions in a cell independent of DNA.
Finally, there is the epigenetic approach, which investigates how environmental factors, such as nutrition and stress, affect FoxO function. This area makes Cheng most excited.
"I'm most interested in the epigenetic aspect of FoxO and autophagy because it is still in its infancy and there is a lot to discover," he said. "Also, environment-induced epigenetic effects on FoxO and autophagy could be reversed via lifestyle modification or interventions. The goal of our research is to discover reliable epigenetic factors that may promote early diagnosis and guide lifestyle interventions to prevent metabolic diseases."
The review article appears in the journal Trends in Endocrinology and Metabolism.
More information: Trends in Endocrinology and Metabolism, 2019. doi.org/10.1016/j.tem.2019.07.009