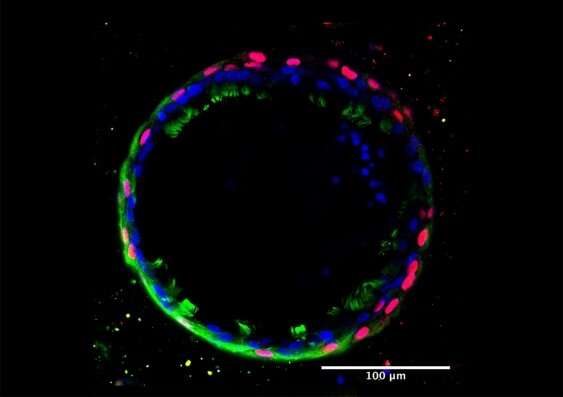
A lung organoid is effectively a patient's avatar that can be used to test whether they would respond to certain CFTR modulator drugs. Credit: miCF Research Centre
UNSW researchers are leading a revolutionary approach to managing treatment of cystic fibrosis using patients' derived lung and gut mini-organs or avatars to test how they will respond to the latest drugs.
UNSW researchers have developed mini gut and lung avatars that could transform the way clinicians treat people with cystic fibrosis (CF), the most common life-limiting genetic disease affecting Australian children.
Only 50 years ago, children with CF were not expected to live beyond their fifth birthday. Today, breakthrough drugs such as CFTR modulators have transformed the outlook for CF patients—enabling some to live more than 50 years—but average life expectancy in Australia still hovers around 35 years.
Genetic focus
Rather than treat the symptoms, CFTR modulators treat the dysfunctional CFTR proteins which are linked to more than 2000 different mutations in the CFTR gene. Because there are so many, it is impossible to use CFTR modulators as a one-size-fits-all solution.
While approved modulators Orkambi, Symdeko and Kalydeco target the more common CFTR mutations to benefit about 60 percent of patients with CF, not all patients show clinical improvement. In addition, the remaining 40 percent of the CF population that have rare CFTR mutations are left without access to treatment.
Compounding the issue is the fact that modulators come at a tremendous cost to the healthcare system—with a cumulative lifetime cost of around $250,000 per patient per year.
New approach
It was these challenges facing the CF respiratory specialists—not being able to target drugs to individuals' specific CFTR mutations, plus the prohibitive costs of the CFTR modulators—that led to collaboration with UNSW researchers. Together they tested patients' stem cell derived mini-organs against various CFTR modulators in a centralised laboratory, the Molecular and Integrative cystic fibrosis (miCF) Research Centre.
Using recent breakthroughs in stem-cell biology, the researchers isolated cells directly from respiratory or gut tissue and encouraged largescale expansion of them to create mini-organs (organoids). Since these organoids were created from the cells of patients with CF, they are effectively an avatar for that person. In other words, if the drug works on their avatar, then it will likely work on the patient. These organoids are cryopreserved in the miCF biobank and can be tested against new drugs in future.
Because the work is carried out in a centralized location that services all of Australia, the miCF Research Centre also ensures the avatar program is cost-effective, as it is not feasible to prepare the avatar cells in every CF center or lab.
National network
The miCF biobank was established by UNSW researchers in the beginning of 2019. It relies on a national network of CF clinicians, scientists and their institutions working together with the miCF Research Centre to co-ordinate tissue procurement from 11 CF clinics across Australia.
Consenting donors with CF provide respiratory and gut tissue at participating sites which is then delivered to the miCF Research Centre. Trained personnel then prepare the organoids using standardized techniques and quality control measures at UNSW's miCF Research Centre.
The miCF research lab uses the organoids to identify drug responsive from non-responsive individuals. Of the patients' avatars identified as responsive to the CFTR modulators, three have been ultra-rare CFTR mutations. The avatar technology effectively acts as a crystal ball that greatly reduces the need for trial and error in targeted CFTR therapy.
The research team is also testing ways to correct the defective CFTR by adding a correct copy of the gene to the cells. These cells serve as an invaluable tool to enhance the current understanding of CF and the translational research efforts that aim to develop new therapeutic agents to fight the disease and shape the future for CF precision medicine in Australia.
Provided by University of New South Wales