In-office gene therapy for wet age-related macular degeneration is coming
Gene therapy is showing promise for one of the most common causes of blindness. Data presented today shows that six patients with wet age-related macular degeneration (AMD) have, so far, gone at least six months without the need for continued injections to control a disease that typically requires treatment every four to six weeks. Researchers say the hope is that gene therapy will free patients from nearly monthly eye injections by offering a potential "one-and-done" treatment. It's not just about convenience; a more consistent treatment may also help people keep more of their vision. The research will be presented today at AAO 2019, the 123rd Annual Meeting of the American Academy of Ophthalmology.
"This is potentially paradigm shifting," said lead researcher, Szilárd Kiss, M.D., director of Clinical Research and chief of the Retina Service in the Department of Ophthalmology at Weill Cornell Medical College in New York City. "It's the next revolutionary leap in treating AMD. When you think about what is science fiction and what is science reality; gene therapy for AMD is becoming a clinical reality."
Dr. Kiss expects a gene therapy for wet AMD could be available within the next three to five years, depending upon the results of further clinical studies and regulatory approvals.
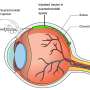
AMD is the most common cause of vision loss and blindness in Americans over age 50, affecting about 2.1 million people nationwide; a number that's expected to explode as the population ages. AMD is a degenerative eye disease that happens when part of the retina is damaged. The damage happens when new, weak blood vessels form behind the retina at the back of the eye. These abnormal vessels leak, causing scaring and killing off the cells that allow us to see.
The first revolutionary leap in AMD treatment occurred a little over a decade ago, with the introduction of a new medication called anti-VEGF therapy. It was the first treatment to inhibit the formation of those damaging, new blood vessels, allowing more than 90 percent of patients to keep their vision, according to clinical trials.
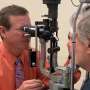
However, in the real world, the actual percentage is closer to 50 percent. One main reason why is that patients are undertreated. This is because most people with AMD must go to the ophthalmologist's office every four to eight weeks for an injection directly into their eye (oftentimes in both eyes). This can be a difficult schedule to maintain for many elderly patients struggling with other maladies and reliant on others to get them to their ophthalmologist visits. It's also unsustainable for the health care system. Last year alone, ophthalmologists performed more than 8 million anti-VEGF injections in the United States.
Researchers have been searching for a better alternative to monthly injections almost from the moment anti-VEGFs were introduced. Gene therapy is emerging as one of the more promising alternatives to long-term anti-VEGF treatment.
The goal of Dr. Kiss' research is to develop a gene therapy that allows the eye to make its own anti-VEGF medicine. The ideal gene therapy would be administered not through a surgical procedure in an operating room, but through an injection into the eye that can be done in the doctor's office, just like routine anti-VEGF treatment is done today.
To do this, Dr. Kiss and colleagues have developed a next-generation vector that can insert into the cells of the eye, genetic material that makes a molecule similar to a widely used anti-VEGF medicine called aflibercept. Once inside the cells, the DNA sequence begins making the aflibercept protein.
"Instead of taking a vile of aflibercept and injecting it into the eye, your eye makes the aflibercept," Dr. Kiss explained. "The goal is a potentially one-and-done treatment. You may need a booster once in a while, but this gene therapy could theoretically last a lifetime."
In animal studies, Dr. Kiss and colleagues have shown that this gene therapy works just as well as an injection of aflibercept, with a favorable and manageable side-effect profile.
Dr. Kiss will be presenting today early data on the first human subjects treated. The phase 1 clinical trial has thus far enrolled 12 patients who received a single injection of gene therapy for wet AMD. Prior to the study, the patients had received an average of 35 anti-VEGF injections; one patient had 109. Since entering the gene therapy trial, the patients required no rescue treatments through the first six months.