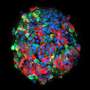
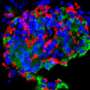
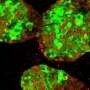
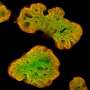
Stem cells could help turn the tide against diabetes, but more research is needed to better control their differentiation and insulin-producing properties. Credit: A*STAR
There's no sugar coating it—diabetes is shaping up to become one of the greatest health challenges in modern times. According to the World Health Organization (WHO), one in eleven individuals suffers from diabetes, and the condition was the direct cause of 1.6 million deaths in 2016, based on the latest publicly available data. WHO has since designated diabetes as one of four priority noncommunicable diseases (alongside cancer, respiratory and cardiovascular diseases) to be addressed by global health authorities.
Closer to home, more than 400,000 Singaporeans live with the diagnosis of diabetes, says the Ministry of Health. This constitutes ten percent of the local disease burden. Meanwhile, a separate study by the National University of Singapore projected that by 2050, Singapore would be home to one million diabetics if current trends continue. Wary of the insidious consequences of unchecked chronic illness, Singapore's Health Minister Gan Kim Yong declared a "war on diabetes," calling for a concerted national effort to prevent disease onset and better manage disease symptoms.
Stem the sugar tide
Despite presenting as a simple disease, diabetes in fact comes in numerous forms. Type I diabetes arises due to an autoimmune reaction that destroys insulin-producing cells called beta cells, located in the pancreas. On the other hand, type II diabetes (T2D) occurs because cells no longer respond to insulin. Lesser known is monogenic diabetes—a rare form of diabetes that is caused by mutations in a single gene.
"Over time, however, pancreatic beta cell failure and beta cell death is a common denominator for all types of diabetes," said Adrian Teo, a Principal Investigator at A*STAR's Institute of Molecular and Cell Biology (IMCB), adding that while obesity is a major contributing factor to diabetes in the West, the main contributing factor in Asia is generally pancreatic beta cell failure. "Although current diabetes medication can help to control blood glucose levels for extended periods of time, they do not cure or even improve pancreatic beta cell health."
This is why Teo's team sees potential in harnessing the power of stem cells to beat back the scourge of diabetes. Unlike most cells in the body, stem cells have the ability to self-renew and can be differentiated into a variety of cell types, including pancreatic beta cells. Hence, stem cells could potentially be used to replace the defunct pancreatic beta cells of diabetic patients, restoring their insulin production and glucose-regulating capabilities.
Rather than rely on embryos as a source of stem cells, Teo envisions obtaining blood cells and fibroblasts—a type of cell in the skin—from diabetic patients, then reprogramming them into human induced pluripotent stem cells (hiPSCs). Subsequently, gene editing may be carried out to correct diabetes-associated mutations or gene variants in these hiPSCs before they are differentiated into pancreatic beta cells and transplanted back into the patient.
"This method potentially allows for the creation of a near-unlimited supply of pancreatic beta cells for cell replacement therapy," said Blaise Su Jun Low, a final year Ph.D. student in Teo's lab. "Because patients will be transplanted with their own cells, graft rejection is less likely to occur."
A different mode of discovery
Beyond cell replacement therapy, hiPSCs can also help shed light on the underlying molecular mechanisms of diabetes. For example, Teo's research group uses hiPSCs from patients diagnosed with maturity-onset diabetes of the young (MODY)—a subtype of monogenic diabetes—to understand how certain gene networks control pancreas and liver development. Both organs are crucial for normal glucose metabolism.
The broad experimental setup is as follows: first, the researchers induce hiPSCs from MODY patients to differentiate into foregut endoderm and human pancreatic precursors—the parts of human embryos that eventually give rise to the pancreas and liver, and then to pancreatic beta-like cells. The team then compares the gene expression pattern of MODY hiPSC-derived foregut endoderm, pancreatic precursors and betalike cells to that of normal individuals.
With this approach, Teo's team discovered that mutations in a gene called HNF4A result in reduced overall expression of genes specifying pancreas and liver development in MODY 1 patients. Importantly, Teo noted that this discovery would not have been possible using mouse models, since mice with one mutant copy of HNF4A do not develop diabetes, unlike the situation in humans.
"Currently, there are more than 14 forms of MODY, each caused by mutations in a different gene (e.g. HNF4A, HNF1A, PAX4 and INS)," Teo explained. "Interestingly, gene variants found in many of these MODY genes are associated with T2D, the most common form of diabetes that affects approximately 90 percent of the diabetic population." The findings from MODY patients may therefore be relevant to the pathophysiology of T2D as well, Teo added.
Furthermore, by using hiPSCs as a platform for genetic screens, researchers may be able to better stratify patients into different treatment groups. At the same time, novel drug targets may be identified based on such screening methods. This brings the ideal of precision medicine for diabetes closer to reality—no longer will a one-size-fits-all solution be applied to all patients, but medicines will be prescribed based on underlying genetic defects that are unique to each diabetic person.
Sweetening the deal
The use of hiPSCs in genetic screening and drug discovery for diabetes is already underway in many labs around the world. On the other hand, therapy involving hiPSC replacement of dysfunctional pancreatic beta cells still has some way to go before it can be approved for use in the clinics.
Low cautioned that protocols for differentiating hiPSCs into pancreatic beta cells are not yet 100 percent efficient, and some residual pluripotent cells may still lurk among differentiated pancreatic beta cells. If these pluripotent cells are also transplanted along with the pancreatic beta cells, they could result in a teratoma—a tumor that could lead to life-threatening complications, she said.
The precise function of hiPSC-derived pancreatic beta cells has yet to be fully verified, Teo continued. "They need to function just like bona fide human pancreatic beta cells or islets," he warned. "Otherwise the glucose levels of the individual will not be properly regulated, posing health risks."
Yet another concern lies with the safety of gene-editing technologies such as the popular CRISPR/Cas 9 system when correcting diabetes-associated gene mutations. Unless undesirable or unexpected off-target consequences of CRISPR-mediated genome editing can be ruled out, the use of gene-edited hiPSCs for cell replacement therapy will likely remain limited, said Teo.
In spite of these challenges, Teo's group remains optimistic and undeterred. "For hiPSC-based cell therapy without any genome editing, there are currently a few clinical trials ongoing to evaluate the ability of these hiPSCs to mature into pancreatic beta cells and regulate blood glucose levels," he said. "Our lab and many others in the world are now working on optimizing the pancreatic differentiation protocol to eliminate residual hiPSCs and improve the functionality of hiPSC-derived pancreatic beta cells such that they behave similarly to bona fide insulin-secreting cells in the islets of the human pancreas."
In fighting the good fight against diabetes, Teo underscored the importance of close collaboration between the lab and clinic. "We believe that partnerships with clinicians and pancreas transplant surgeons are a win-win model that can help us translate our scientific efforts toward eventual therapeutic value for our patients and society," he emphasized.
More information: Natasha Hui Jin Ng et al. HNF4A Haploinsufficiency in MODY1 Abrogates Liver and Pancreas Differentiation from Patient-Derived Induced Pluripotent Stem Cells, iScience (2019). DOI: 10.1016/j.isci.2019.05.032