Credit: Elena Hartley/Boston Children's Hospital
A pilot gene therapy treatment for sickle cell disease, restoring patients' ability to make fetal hemoglobin, has produced good results in the first three patients to receive it. Investigators at Boston Children's Hospital reported the findings of their ongoing clinical trial this week at the American Society of Hematology (ASH) annual meeting.
The three adult patients, all treated at Boston Children's, have now been followed for 8, 10, and 18 months, respectively. None have shown adverse events from the treatment. And all indicators suggest that the gene therapy has "taken" and that sickling of red blood cells has been reduced. The trial has now begun to enroll adolescents with sickle cell disease.
Restoring fetal hemoglobin
The gene therapy, drawing upon some 70 years of research, is designed to get people to make a fetal form of hemoglobin, normally not made after birth. The mutations causing sickle cell disease don't affect fetal hemoglobin, so the red blood cells that carry it would be expected not to sickle. The treatment silences BCL11A, the gene that shuts down fetal hemoglobin production. In so doing, both restarts fetal hemoglobin production and suppresses production of the adult, sickling form of hemoglobin.
Tests on the three patients showed that blood stem cells modified with the gene therapy vector (a genetically engineered virus with advanced safety features) successfully engrafted in patients' bone marrow and persisted in their blood circulation. As hoped, levels of BCL11A protein were significantly reduced in patients' red blood cell precursors, but remained normal in other blood cells.
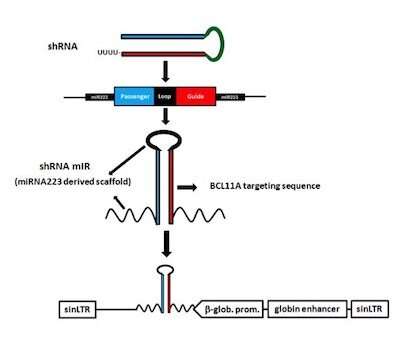
The gene therapy vector incorporates a short hairpin RNA (shRNA) that inactivates the BCL11A gene and is embedded in a microRNA to get it into cells. To make this assembly work in the right place at the right time, the team hooked it to a promoter of beta hemoglobin expression, together with regulatory elements active only in red blood cell precursors. Credit: Children's Hospital Boston
Just as the researchers had hoped, all three patients showed a significant increase in fetal hemoglobin. One continued to get red-blood-cell transfusions because of pre-existing blood vessel damage in the brain, a known complication of sickle cell disease. But in the other two patients, red blood cells containing fetal hemoglobin made up about 70 percent of all red blood cells. In both, fetal hemoglobin levels were thought to be high enough to prevent sickling. In addition, the patients no longer show the anemia associated with sickle cell disease.
"The gene therapy treatment has been safe and well tolerated," says Erica Esrick, MD, co-principal investigator on the clinical trial and a pediatric hematologist at Boston Children's. "We are pleased to see a substantial and stable increase in total fetal hemoglobin and the amount of fetal hemoglobin per cell."
Designing a sickle cell gene therapy vector
It's been known since the 1970s that some people with the sickle cell mutation keep producing fetal hemoglobin and have milder disease. Vijay Sankaran, MD, Ph.D. and Stuart Orkin, MD of Boston Children's zeroed in on BCL11A in 2008. In a 2011 study, Orkin's team suppressed BCL11A in a mouse model and successfully reversed sickle cell disease.
Manny with Esrick. Credit: Sam Ogden/Dana-Farber Cancer Institute
For the clinical trial, a team led by David A. Williams, MD, Boston Children's Chief Scientific Officer, went on to engineer an optimized gene therapy vector. It suppresses BCL11A, but only in precursors of red blood cells.
"Our approach reverses the physiologic hemoglobin switch to simultaneously increase fetal hemoglobin and directly reduce sickling hemoglobin," says Williams, who is also the sponsor of the clinical trial. "Other trials are adding genes that encode fetal hemoglobin or corrected, non-sickling adult hemoglobin, without directly decreasing expression of the sickle hemoglobin gene. We predict this strategy is a more efficient and effective way to reduce or even eliminate the sickling of cells."
Manny's story
Manny Johnson of Boston, 22, was the first patient treated; his early results were presented at ASH last year. His sickle cell disease was severe and had required him to have monthly blood transfusions. Now, more than 18 months later, he's remained symptom-free and has not needed transfusions.
Now that the gene therapy appears safe in adults, the clinical trial has gone on to treat its first adolescent patients. It has also begun enrolling children younger than 12.
Provided by Children's Hospital Boston