Indirect effects of SARS-CoV-2 on the kidneys: beware of the genetic background
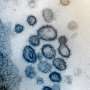
SARS-CoV-2-associated acute kidney injury is not uncommon. Many hospitalized COVID-19 patients—as many as 80% of critically ill COVID-19 patients, according to a current study—develop acute kidney injury, but without requiring dialysis in most cases. Proteinuria and hematuria are present in about half the cases. Examinations of renal tissue from deceased patients show injury to the renal corpuscles (glomeruli) or filter membrane (podocytes), and to the renal tubules (tubular cells). Given that SARS-CoV-2 uses the ACE2 receptor to infect cells, and that tubular cells and podocytes express ACE2, it is clear that the kidney can be a specific tissue targeted by the virus.
The current study, published in Clinical Kidney Journal (CKJ), reports on two COVID-19 patients with injury to both these renal structures. Both patients were over 50 years of age and were known to have high blood pressure; one had cardiac insufficiency, the other hepatitis B. Both had been complaining of coughing for four days; one of them also had a fever. On admission, they had pulmonary CT findings typical of COVID-19 and differently elevated creatinine levels indicating reduced renal function (acute kidney injury). Within a few days, one patient required ventilation, whereas the other only needed oxygen; dialysis was not required in either case. Both recovered their pulmonary function within one to two weeks (extubation on day 14). Kidney function also improved, but they both had persistent, high proteinuria. Kidney biopsies were therefore performed. These showed capillary collapse in the glomeruli ("collapsing glomerulopathy/FSGS"), podocyte swelling and glomerular deposits of immunoglobulins (IgM and complement factor C3). Tubule injury (tubulointerstitial lesions), partly with tubule atrophy and necrosis, as well as inflammatory cells (monocytes, macrophages) were also found.
Although both patients had positive virus detection from the throat swab, SARS-CoV-2 was not detected in blood, urine or in kidney tissue, despite highly sensitive RT-PCR testing. Molecular genetic analysis showed variants in the APOL1 gene (homozygous G1 polymorphism and G1/G2 heterozygosity), which are known to be associated with a predisposition (increased risk) for kidney disease.
"The histological findings and the negative PCR results do not definitively rule out direct viral infection of the kidneys, but it seems probable that the injury was not in fact caused directly by the viral infection, but rather by a SARS-CoV-2-induced inflammatory reaction," explains the study's corresponding author, Professor Ziad Massy, Paris/France. "Particularly in the context of genetic APOL1 risk variants, SARS-CoV-2 might have triggered the 'collapsing FSGS' in accordance with the 'second hit' hypothesis."
"SARS-CoV-2 has meanwhile arrived in every country in the world. This means that we will be confronted very often with SARS-CoV-2-induced kidney injury in those regions where the APOL1 gene variants G1/G2 are common," added Professor Alberto Ortiz, Editor-in-Chief of the CKJ. "People of African-American descent, in particular, often express APOL1 risk variants, so in addition to lung problems, renal complications and associated conditions, especially, can be expected in the U.S.A., among other countries."
More information: Aymeric Couturier et al, Indirect effects of severe acute respiratory syndrome coronavirus 2 on the kidney in coronavirus disease patients, Clinical Kidney Journal (2020). DOI: 10.1093/ckj/sfaa088