Mutations linked to intellectual disability point to overly active ion channel
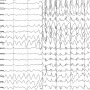
Two mutations identified in individuals with developmental and epileptic brain disease can be traced back to the same ion channel. Researchers have now elucidated how both independent mutations affect the channel's function: by making it overly active and highly sensitive to stimulation. The findings are an important step towards unraveling what causes the patients' symptoms.
Developmental and epileptic encephalopathies are a heterogeneous group of disorders characterized by epilepsy and intellectual disability. Genetic analysis pointed to the involvement of an ion channel called TRPM3. This channel is activated by heat and a variety of chemical ligands. It helps us detect noxious heat, and plays an important role in inflammatory pain.
"Two different mutations in the gene encoding TRPM3 were identified in nine individuals with a diagnosed developmental and epileptic encephalopathy," explains Thomas Voets (VIB-KU Leuven) whose research is focused on ion channels and their role in pain and heat sensation. "Since the functional consequences of the mutations remained elusive, we set out to understand how disturbances of this particular ion channel can cause intellectual disability and epileptic seizures."
An overly active channel
Voets, together with his colleague Joris Vriens (KU Leuven) and their team at the Laboratory for Ion Channel Research, could show that both mutations affect TRPM3 channel gating.
"Clinical reports pointed out that next to the seizures and intellectual disability, several of the affected patients showed altered sensitivity to heat and pain, which hinted at a modified altered function of TRPM3," says Vriens.
The team could indeed demonstrate that both mutations lead to a significant gain in channel activity. Not only was basal activity increased, they also observed a higher sensitivity to stimulation by an endogenous neurosteroid and heat. Most strikingly, one of the two mutations resulted in even more pronounced functional alterations, including anomalous activation and reduced sensitivity to an anticonvulsant drug.
"The two mutations identified in the TRPM3 gene give rise to channels with substantially altered functional properties. Whereas the individual effects of both mutations differ, both can be considered as strong gain-of-function mutants, with increased activity, both under basal conditions and when stimulated," says Voets.
"These two mutations also provide the first genetic evidence that link TRPM3 to a pain phenotype in humans," adds Vriens. The researchers propose that this increased channel activity may lie at the basis of seizure development and neurodevelopmental symptoms in patients.
More information: Evelien Van Hoeymissen et al. Gain of channel function and modified gating properties in TRPM3 mutants causing intellectual disability and epilepsy, eLife (2020). DOI: 10.7554/eLife.57190