Droplet biosensing method opens the door for faster identification of COVID-19
Mechanical engineering associate professor Jiangtao Cheng and electrical and computer engineering assistant professor Wei Zhou have developed an ultrasensitive biosensing method that could dramatically shorten the amount of time required to verify the presence of the COVID-19 virus in a sample. Their peer-reviewed research was published in ACS Nano on June 29.
There's significant room to improve the pace of coronavirus testing, Cheng and Zhou have found. Current COVID-19 verification tests require a few hours to complete, as verification of the presence of the virus requires the extraction and comparison of viral genetic material, a time-intensive process requiring a series of steps. The amount of virus in a sampling is also subject to error, and patients who have had the virus for a shorter period of time may test negative because there is not enough of the virus present to trigger a positive result.
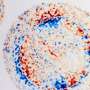
In Cheng and Zhou's method, all of the contents of a sampling droplet can be detected, and there is no extraction or other tedious procedures. The contents of a microdroplet are condensed and characterized in minutes, drastically reducing the error margin and giving a clear picture of the materials present.
The key to this method is in creating a surface over which water containing the sample travels in different ways. On surfaces where drops of water may "stick" or "glide," the determining factor is friction. Surfaces that introduce more friction cause water droplets to stop, whereas less friction causes water droplets to glide over the surface uninhibited.
The method starts by placing a collected sample into liquid. The liquid is then introduced into an engineered substrate surface with both high and low friction regions. Droplets containing sample will move more quickly in some areas but anchor in other locations thanks to a nanoantenna coating that introduces more friction. These stop-and-go waterslides allow water droplets to be directed and transported in a programmable and reconfigurable fashion. The "stopped" locations are very small because of an intricately placed coating of carbon nanotubes on etched micropillars.
These prescribed spots with nanoantennae are established as active sensors. Cheng and Zhou's group heats the substrate surface so that the anchored water droplet starts to evaporate. In comparison with natural evaporation, this so-called partial Leidenfrost-assisted evaporation provides a levitating force which causes the contents of the droplet to float toward the nanoantenna as the liquid evaporates. The bundle of sample particles shrinks toward the constrained center of the droplet base, resulting in a rapidly-produced package of analyte molecules.
For fast sensing and analysis of these molecules, a laser beam is directed onto the spot with the packed-in molecules to generate their vibrational fingerprint light signals, a description of the molecules expressed in waveforms. This method of laser-enabled feedback is called surface-enhanced Raman spectroscopy.
All of this happens in just a few minutes, and the fingerprint spectrum and frequency of the coronavirus can be quickly picked out of a lineup of the returned data.
Professor Cheng and Zhou's team is pursuing a patent on the method, and are also pursuing funding from the National Institutes for Health to deliver the method for widespread use.
More information: Junyeob Song et al. Partial Leidenfrost Evaporation-Assisted Ultrasensitive Surface-Enhanced Raman Spectroscopy in a Janus Water Droplet on Hierarchical Plasmonic Micro-/Nanostructures, ACS Nano (2020). DOI: 10.1021/acsnano.0c04239