Modulating bone cell recruitment to prevent osteoporosis
NUS biologists have identified a pathway to reduce bone resorption and maintain bone health for potentially better treatment outcomes.
Osteoporosis is the most common age-related bone disease which affects hundreds of millions of individuals worldwide. It is estimated that one in three women and one in five men aged over 50 suffer from osteoporotic bone fractures. In aging populations, the incidence of hip fractures increases dramatically, resulting in a high risk of morbidity and mortality. Hence, osteoporosis is an immediate and significant health concern in Singapore and worldwide.
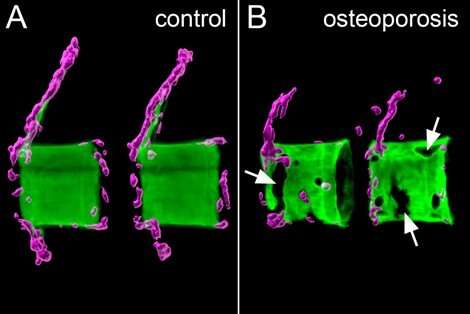
Osteoporosis is caused by excessive activity of bone resorbing cells, called osteoclasts, while activity of bone-forming cells, called osteoblasts, is reduced. In healthy individuals, a balanced activity of these two cell types allows constant bone turnover to maintain healthy and strong bones. In osteoporosis, disproportionate bone resorption leads to low bone mineral density and consequently weak and fracture-prone bones. When new bone formation is unable to catch up with bone loss, bone eventually weakens, and becomes more prone to fractures.
Most current osteoporosis therapies include the use of bisphosphonates, which block osteoclast activity and thus prevent excessive bone resorption. However, prolonged treatment with these drugs eliminates the necessary bone turn-over leading to increased fracture risk and other unwanted side effects. Therefore, there is an urgent need to develop new strategies that overcome the limitations of current treatments.

A research team led by Prof Christoph Winkler and comprising his colleagues Dr. Phan Quang Tien and Dr. Tan Wen Hui from the Department of Biological Sciences, NUS, in collaboration with groups at the Genome Institute Singapore (GIS) and the University of Wuerzburg has identified a mechanism to control the recruitment of osteoclasts to sites of bone resorption. Using genetic analysis in a small laboratory fish model, the Japanese medaka (Oryzias latipes), the research team identified a small protein, the chemokine CXCL9, that is released from osteoblasts located on the surface of the bone matrix. Under osteoporotic conditions, CXCL9 diffuses towards reservoirs that hold osteoclast precursors. These osteoclast precursors produce a receptor, CXCR3, on their cell surface. Upon activation by CXCL9, the osteoclast precursors are mobilized and migrate long distances in a highly directed fashion towards the bone matrix, where they start resorbing bone.
Both CXCL9 and its receptor CXCR3 have long been known to modulate the migration of immune cells to inflammation sites, for example in psoriasis and rheumatoid arthritis. Their role in osteoporosis and recruitment of bone cells, however, is unknown. There are several chemical inhibitors blocking CXCR3 activity that have had little success in clinical tests for the treatment of psoriasis. The research team showed that these inhibitors are highly effective in blocking osteoclast recruitment and protecting bone from osteoporotic insult.
Prof Winkler said, "Our studies provided new avenues to osteoporosis therapy. The new strategy allows a fine-tuned modulation of osteoclast numbers that are recruited to bone matrix rather than a widespread blockage of osteoclast activity as in traditional therapies. This has major advantages as excessive bone resorption can be prevented in a targeted manner but normal bone turnover will still continue. This offers potential to avoid increased fracture risks in osteoporosis patients and to maintain healthy bone for improved quality of life."
More information: Quang Tien Phan et al. Cxcl9l and Cxcr3.2 regulate recruitment of osteoclast progenitors to bone matrix in a medaka osteoporosis model, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.2006093117