Non-Hodgkin lymphoma treatment gets the green light for use in England
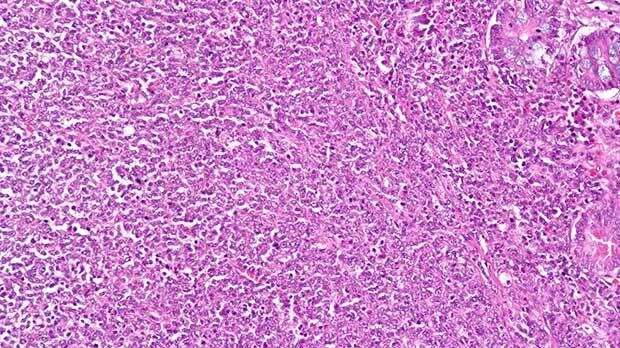
The National Institute for Health and Care Excellence (NICE) has recommended a new combination therapy for adults with an aggressive type of non-Hodgkin lymphoma on the NHS in England.
After an initial rejection back in March, a combination of polatuzumab vedotin (Polivy) and two existing cancer drugs—rituximab and bendamustine—will now be available to some adults with diffuse large B cell lymphoma. It will be a new option for people whose cancer has either not responded to, or come back after, initial treatment, and who are unable to have a stem cell transplant.
NICE decisions are usually adopted in Wales and Northern Ireland as well as England, so the drug should now be available to patients in all three nations. Scotland is reviewing polatuzumab vedotin for NHS use in a separate process.
A new treatment option
Around 4,800 people in England have diffuse large B cell lymphoma and according to NICE, around half of these cancers do not respond to, or come back, following initial treatment.
If someone isn't fit enough to have intensive therapy—including a stem cell transplant—at this point, there is no standard of care. This includes many people who are older, or who have already had a stem cell transplant. It also includes those whose disease is not controlled enough to be eligible for a stem cell transplant.
But this decision means a further treatment option is now available for eligible patients in these groups. NICE estimates around 530 people will be eligible for the treatment, which combines polatuzumab vedotin, an antibody drug that helps to deliver chemotherapy directly to cancer cells, with the targeted cancer drug rituximab and the chemotherapy drug bendamustine.
Updated data
In clinical trials, this three-drug combo was compared to the combination of just rituximab and bendamustine, which NICE agreed is a reasonable comparison for what patients might already receive on the NHS.
Inital trial results found more people responded to the combination, which also increased the time before the disease spreads significantly and improved survival.
"Evidence from clinical trials shows this drug combination could be effective for many blood cancer patients, although it's not yet clear whether it could help them stay cancer-free in the longer-term," said Samuel.
This uncertainty led to NICE rejecting the treatment in March. But since then the company has submitted updated clinical trial data, as well as a new price. Based on this additional information, NICE agreed that the treatment was a plausibly cost-effective use of NHS resources.
The treatment also benefitted from meeting NICE's criteria to be a life-extending treatment for patients at the end of life, allowing NICE extra flexibility to give it the green light for NHS use.
"Polatuzumab vedotin, in combination with other treatments, is a significant step forward in the treatment of lymphoma, giving patients a greater chance of survival, which is why the NHS has worked closely with NICE to reach a deal that not only benefits patients but is also a fair price for taxpayers," says John Stewart, NHS director of specialized commissioning.
Samuel added that it was great for patients that NICE, NHS England and the treatment's manufacturer have been able to work together to agree a deal and make the treatment available.
More information: Polatuzumab vedotin with rituximab and bendamustine for treating relapsed or refractory diffuse large B-cell lymphoma: www.nice.org.uk/guidance/gid-t … termination-document