Model maps out 'arms race' between cancer and immune system, predicts effectiveness of immunotherapy
Researchers have created a mathematical model that can determine the impact of the immune system on tumor evolution. The information gained from using this model may be able to be used to predict whether immunotherapy is likely to be effective for a patient's cancer, helping to guide treatment decisions.
Immune cells recognize tumors by detecting molecules present on the surface of cancer cells, known as neoantigens. Neoantigens are "faulty" molecules that are made as the result of changes to the genetic code (mutations) within cancer cells. When immune cells scan the body, they recognize these faulty molecules as being non-self, which triggers an immune response against the cancer.
In order to overcome destruction by the immune system, the cancer cells fight back by developing "cloaking" mechanisms which they can use to hide from immune cells. Consequently, the cancer can continue to grow undetected within the body.
The team, led by Dr. Eszter Lakatos and Professor Trevor Graham from Barts Cancer Institute, and Professor Andrea Sottoriva, Team Leader in Evolutionary Genomics and Modeling at The Institute of Cancer Research, London, set out to develop a computational model to map the arms race that occurs between cancer cells and immune cells as tumors evolve.
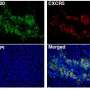
Using genomic data from bowel, stomach and endometrial cancers available from the Cancer Genome Atlas, the team developed a model that works out the interplay between the immune system and cancer cells using the genetic codes of cancer cells as its input.
By calculating the number of neoantigens present in a tumor and looking at how fast they have accumulated, the model is able to predict when the cancer is likely to activate its cloaking mechanisms against the immune system.
Modeling to predict how cancers will evolve
Dr. Lakatos said, "Mathematical modeling can help us re-create biological processes even when only a small part of the picture is available. It is especially useful in determining how a cancer developed, as we typically only see a single snapshot of that process. We developed our model to characterize all possible developing cancers—including the ones that are successfully destroyed by the immune system—to highlight what measurements are most informative about their future evolution."
The research was published today in Nature Genetics and primarily funded by Cancer Research UK, with additional support from Wellcome and the National Institutes of Health.
The predictions from the model may have important implications for immunotherapy sensitivity in patients with cancer. Immunotherapies are a group of drugs which boost the activity of the patient's immune system to help fight their cancer.
Such drugs have been shown to be extremely effective against some cancer types, even achieving cure in some cases. However, this is not the case for all patients and not all tumors respond to immunotherapy. Being able to know ahead of time if immunotherapy is likely to be effective would be extremely valuable for patient management.
A step toward understanding the interplay between immune and tumor cells
Professor Graham said, "Boosting the immune system is an incredibly effective way to treat cancer, but it has been difficult to understand why some patients respond to immunotherapy and others don't. Our study is a step toward understanding the interplay between immune cells and tumor cells, and we hope that this understanding might prove useful for guiding treatment decisions in the future."
The new mathematical model may help researchers make predictions about people's cancers, informing on whether immunotherapy is likely to be an effective treatment option for a patient based on the cancer cells' immune cloaking status.
The model predicts that immunotherapy may be most effective when it is administered after a cancer has cloaked itself, as at this point the therapy could be used to reinvigorate the immune system to recognize and fight the cancer.
The model also predicts that if the cancer has not activated immune cloaking mechanisms early on in its development, immunotherapy may be effective initially but the long-term response to the treatment may not be so successful.
Professor Sottoriva said, "Our model helps us understand, through mathematics, the arms race that takes place within the body between a tumor and the patient's immune system. Cancers are constantly adapting and evolving—and can often dodge the effects of treatment or hide themselves from the immune system. Our study gives us a valuable tool for understanding and predicting how cancers will evolve and interact with the immune system, so we can anticipate cancer's next move, and devise new treatment strategies for patients."
The team has developed a model that expands the understanding of the action of the immune system on a tumor just from looking at the genetic codes of cancers. Because genetic analysis is becoming routine in the NHS, knowing how to use the genetic data to select patients for treatment and improve understanding of why immunotherapies are less effective in some patients than in others is very useful.
The team is now keen to apply their model to cancers that have been treated with immunotherapy to see if the predictions are accurate.
More information: Eszter Lakatos et al. Evolutionary dynamics of neoantigens in growing tumors, Nature Genetics (2020). DOI: 10.1038/s41588-020-0687-1