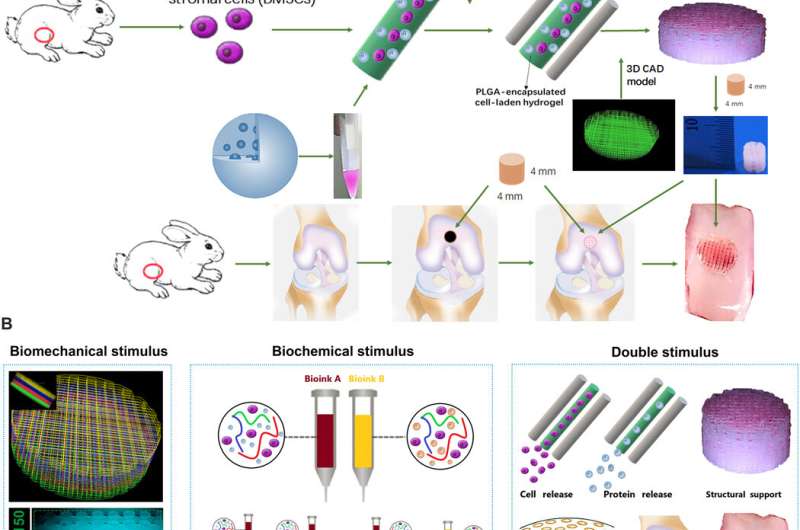
Schematic presentation of the study design and scaffold construction. (A) Schematic Illustration of the study design with 3D bioprinted dual-factor releasing and gradient-structured MSC-laden constructs for articular cartilage regeneration in rabbits. Schematic diagram of construction of the anisotropic cartilage scaffold and study design. (B) A computer-aided design (CAD) model was used to design the four-layer gradient PCL scaffolding structure to offer BMS for anisotropic chondrogenic differentiation and nutrient supply in deep layers (left). Gradient anisotropic cartilage scaffold was constructed by one-step 3D bioprinting gradient polymeric scaffolding structure and dual protein-releasing composite hydrogels with bioinks encapsulating BMSCs with BMP4 or TGFβ3 μS as BCS for chondrogenesis (middle). The anisotropic cartilage construct provides structural support and sustained release of BMSCs and differentiative proteins for biomimetic regeneration of the anisotropic articular cartilage when transplanted in the animal model (right). Different components in the diagram are depicted at the bottom. HA, hyaluronic acid. Credit: Science Advances, doi: 10.1126/sciadv.aay1422
Cartilage injury is a common cause of joint dysfunction and existing joint prostheses cannot remodel with host joint tissue. However, it is challenging to develop large-scale biomimetic anisotropic constructs that structurally mimic native cartilage. In a new report on Science Advances, Ye Sun and a team of scientists in orthopedics, translational research and polymer science in China, detailed anisotropic cartilage regeneration using three-dimensional (3-D) bioprinting dual-factor releasing gradient-structured constructs. The team used the dual-growth-factor releasing mesenchymal stem cell (MSC)-laden hydrogels for chondrogenic differentiation (cartilage development). The 3-D bioprinted cartilage constructs showed whole-layer integrity, lubrication of superficial layers and nutrient supply into deeper layers. The scientists tested the cartilage tissue in the lab and in animal models to show tissue maturation and organization for translation to humans after sufficient experimental studies. The one-step, 3-D printed dual-factor releasing gradient-structured cartilage constructs can assist regeneration of MSC- and 3-D bioprinted therapy for injured or degenerative joints.
Chondrogenesis in the lab
Articular cartilage typically forms an elastic connective tissue in the joint. Cartilage injury is an extremely common impairment although with limited self-healing capacity due to the low cellularity and avascular nature of the tissue. Damage to cartilage can be debilitating and cartilage or joint reconstruction is currently considerably challenging in the research lab. In clinical practice arthritic joints can be replaced by total joint arthroplasty using metallic and synthetic prosthesis. However, the existing joint prostheses cannot remodel (or integrate) with host tissue, which can lead to long-term functional impairments that can only be addressed via biological regeneration of the joint. Scientists have recently developed mesenchymal stem cell (MSC) transplants to stimulate directional differentiation into chondrocytes as a new method for cartilage repair. However, it is still challenging to mimic the gradient anisotropic structure and signaling approaches in different layers to induce zonal-dependent chondrogenic differentiation and extracellular matrix (ECM) deposition to promote osteochondral regeneration. In this work, Sun et al. developed a 3-D bioprinted, dual-factor releasing, gradient-structured MSC-laden construct to implant and establish whole-layer cartilage regeneration in an animal model.
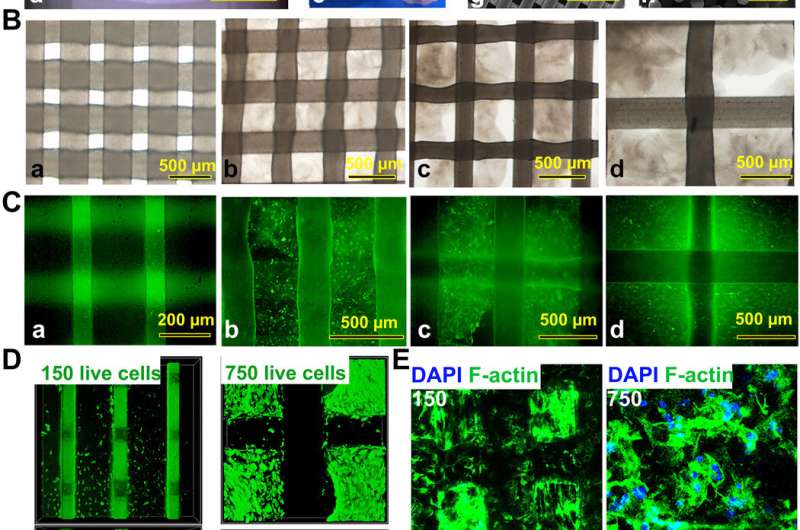
3D bioprinted gradient cartilage scaffold for implantation. (A) Gross appearance of (a) human-scale and (b and c) rabbit-scale cartilage scaffold (b, NG with 150-μm spacing; c, NG with 750-μm spacing). Top view of the rabbit cartilage scaffold is also shown (d, NG with 150-μm spacing; e, NG with 750-μm spacing; f, gradient scaffold with 150- to 750-μm spacing) atop of the SEM images (g, horizontal section; h, vertical section) taken for the 150-μm NG scaffold to demonstrate the precise alignment of the PCL fibers in the printed scaffold. (B) Deconstruction of the gradient scaffold. The structure of the gradient scaffold was deconstructed into four layers. Microscopic appearance of the hydrogel-PCL composite structure in each layer demonstrated good interconnectivity and delicate, orderly aligned structure for each layer. (C and D) Good cell viability is shown respectively for superficial and deep layers after printing with live/dead assay (green, live cells; red, dead cells) (C) under a microscope and (D) under a confocal microscope. DAPI, 4′,6-diamidino-2-phenylindole. (E) Cell spreading in superficial and deep layers with cytoskeleton staining. (F) Immunostaining for cartilage markers in superficial and deep layers. Expression of COL2A1 and PRG4, the lubrication markers, was significantly higher in the superficial layers with small pore size (a and b), while the chondrogenic cells in the deep layers (c and d) mostly presented with hypertrophic phenotype (COL10A1 expression). Photo credit: Ye Sun, First Affiliated Hospital of Nanjing Medical University. Credit: Science Advances, doi: 10.1126/sciadv.aay1422
3-D bioprinting cartilage constructs
The team used 3-D bioprinting to develop different joint tissue constructs for joint reconstruction. They mimicked the native cartilage by including biochemical stimulus (BCS) with diverse growth factor releasing constructs and biomechanical stimulus (BMS) with small pore sizes to induce chondrogenesis. They then created a third cartilage construct as a double stimulus (DS) group to include the two versions of stimuli. For growth factors, the team chose a combination of bone morphogenetic protein (BMP4) and the transforming growth factor β3 (TGFβ3) in the cartilage construct to regenerate complex inhomogeneous joint tissues. Sun et al. then developed a hydrogel to deliver the growth factors and used poly(lactic-co-glycolic acid) (PLGA) microspheres as a carrier/vehicle. The team maintained constant fiber spacing for the BMS (biomechanical stimulus) group and BCS (biochemical stimulus) group to develop the non-gradient scaffolds, while introducing gradually varying fiber spacing for scaffolds in the DS (double stimulus) group. The scientists also used poly(ε-caprolactone) (PCL) polymers and integrated them to the biomimetic scaffold construct. In this way, they developed rabbit cartilage constructs using 4 x 4 x 4 mm scaffolds and human cartilage constructs using 14 x 14 x 14 mm scaffolds.
The 3D printing process using OPUS system showing the hydrogel-PCL patterning and gradient microchannels of every layer and the whole cartilage construct. Credit: Science Advances, doi: 10.1126/sciadv.aay1422
Testing the effects of the scaffolds and cartilaginous matrix formation in the lab
To test the impact of the growth factors on bone marrow stromal cell (BMSC) viability and proliferation, Sun et al. cultured BMSCs in hydrogels for seven days. The microspheres first released sustained and controlled volumes of growth factors in the lab. Then the scientists followed the experiment with cell viability and proliferation assays on printed scaffolds to note the survival of BMSCs at 60 minutes (day zero), seven days and 21 days after bioprinting. The live cells showed increased cell viability on day zero, followed by sustained growth from days three to 21. After 21 days, Sun et al. noted good 3-D cell anchorage on the scaffold and the work showed the development of a favorable microenvironment for BMSC growth and differentiation to form chondrocytes in the lab.
Before translating the 3-D bioprinted scaffold into an animal model, Sun et al. tested if the delivery of growth factors could induce layer-specific BMSC differentiation into chondrocytes. They followed an experimental protocol and observed aggrecan (a glial marker protein) and type II collagen to form hyaline articular chondrocyte-like cells. The team then transplanted the cartilage scaffolds into an animal model and examined their functionality for 12 weeks in vivo. The enhanced mechanical properties of the resulting articular cartilage showed promising regeneration to provide structural support for the newly formed cartilage tissue. After 12 weeks, Sun et al. conducted immunofluorescence imaging to show resemblance of the implanted cartilage constructs to native joint cartilage surrounded by the cartilage matrix.
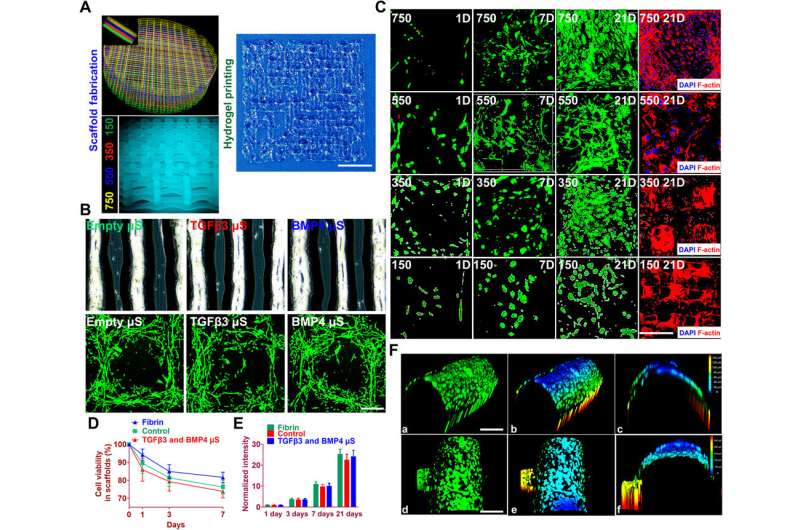
Cell viability and anchoring in the printed anisotropic scaffold. (A) Schematic of anisotropic cartilage scaffold construction with fabrication of gradient scaffolding structure (left) and large-scale printing of aligned protein-releasing BMSC-laden hydrogel (right). Scale bar, 1 mm. (B) Gross appearance of PLGA μS–encapsulated BMSC-laden hydrogel under a microscope (top). Printed cell-laden hydrogel causes cell alignment in a longitudinal direction of the printed paths, forming a reticular network with cell interaction (bottom). (C) Live/dead cell assays showed ≥95% cell viability maintained through day 1 to 21 for all four layers with gradient spacing (4th row, 150-μm spacing; 3rd row, 350-μm spacing; 2nd row, 550-μm spacing; 1st row, 750-μm spacing). Immunostaining of cytoskeleton (rightmost column) showed cell spreading both in the hydrogel and on the PCL fibers throughout the four layers of the construct. Scale bar, 500 μm. (D and E) Quantified cell viability and proliferation in the printed scaffolds. (F) Cell anchoring in the scaffolds. (a to c) At day 21, good 3D anchoring to the PCL fiber cylinder was observed for the MSC cells released from the hydrogel. (d to f) Similar cell anchoring was observed for PCL fibers in adjacent layers. (b), (c), (e), and (f) are 3D demonstration of cell anchoring in (a) and (d), respectively. Scale bars, 100 μm. Photo credit: Ye Sun, First Affiliated Hospital of Nanjing Medical University. Credit: Science Advances, doi: 10.1126/sciadv.aay1422
Better repairing effects of cartilage implants in a rabbit knee cartilage defect model
Sun et al. used rabbit experimental models to test the capacity of cartilage scaffolds during knee repair. They constructed the scaffolds using one-step 3-D bioprinting to provide structural support and sustained release of cells. The experiment facilitated biomimetic regeneration of the native articular cartilage and the implant showed better integration at the site of defect by 24 weeks for the dual stimuli (DS) experimental group compared with both BMS and BCS groups. The team monitored the site of transplantation with magnetic resonance imaging (MRI) for notable healing of the articular cartilage after 24 weeks (within six months). The results showed better cartilage repair and joint management with the DS cohort compared to the BCS or BMS animal groups.
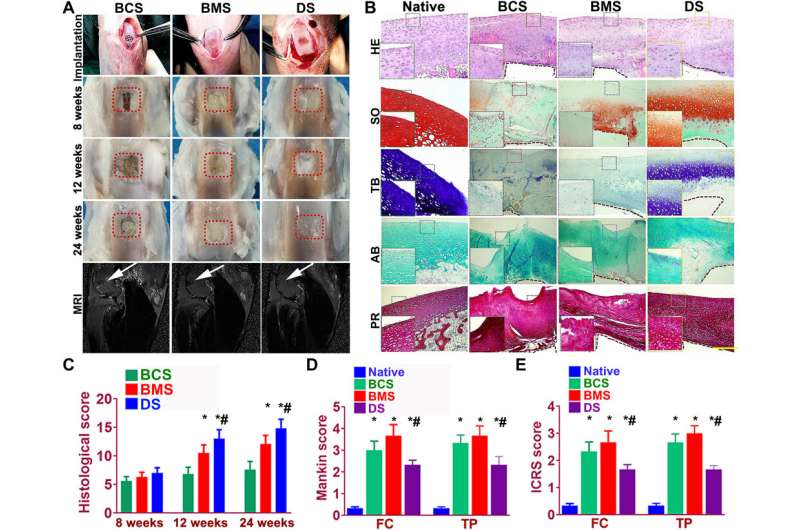
Dual-factor releasing, and gradient-structured cartilage scaffold demonstrated better repairing effect of anisotropic cartilage in rabbit knee cartilage defect model in vivo. (A) Scaffold implantation process and gross appearance of the repair cartilage at 8, 12, and 24 weeks. MRI was made for the operated knee joint (fifth row), demonstrating significant better resolution of subchondral edema and healing of the articular surface (white arrowheads) for joint transplanted with DS scaffolds. (B to F) Chondroprotective effects of the scaffolds were compared by (B) histological scoring evaluation of the repaired cartilage tissue during in vivo implantation. (C) Mankin score and (D) ICRS (International Cartilage Repair Society) histological score of articular cartilage in the femoral condyle (FC) and tibial plateau (TP) in both groups with scaffold implantation. *P < 0.05 between the native group and other groups. #P < 0.05 between the BCS group and the DS group. Data are presented as averages ± SD (N = 6). (A) Histomorphological analysis of the neocartilage tissue at 24 weeks. PR, picrosirius red. The left bottom panels are higher-resolution pictures of the formed neocartilage outline in the colored square boxes. (a to e) Sections were stained with (a) H&E, (b) Safranin O, (c) TB, and (d) AB staining to indicate the presence of proteoglycans in different groups compared with native cartilage. (e) Picrosirius red was used to stain collagens I and III. The brown irregular area at the interface under the formed neocartilage was undegraded PCL material as supporting structure for the scaffolds. Photo credit: Ye Sun, First Affiliated Hospital of Nanjing Medical University. Credit: Science Advances, doi: 10.1126/sciadv.aay1422
The team observed articular cartilage transitions from the superficial zone to deeper zones and tested the properties of the generated cartilage comparatively with the native cartilage. As before, the DS-scaffold showed ingrown microvessels with improved growth compared to the other groups (BCS and BMS). In this way, Ye Sun and colleagues generated 3-D bioprinted anisotropic constructs with structural integrity for joint reconstruction, articular cartilage regeneration and functional knee articular cartilage construction in a rabbit model. The 3-D bioprinted functional constructs acted as prosthesis during joint replacement or cartilage repair to heal injured or degenerate joints in animal models. The scientists will conduct further experiments to understand the functional sustenance of joint construction in animal models. After further experiments in pre-clinical animal models, the team envision translating the 3-D bioprinted constructs via mini-invasive arthroscopy to replace damaged or degenerative joints within humans.
More information: Ye Sun et al. 3D bioprinting dual-factor releasing and gradient-structured constructs ready to implant for anisotropic cartilage regeneration, Science Advances (2020). DOI: 10.1126/sciadv.aay1422
Chang H Lee et al. Regeneration of the articular surface of the rabbit synovial joint by cell homing: a proof of concept study, The Lancet (2010). DOI: 10.1016/S0140-6736(10)60668-X
April M Craft et al. Generation of articular chondrocytes from human pluripotent stem cells, Nature Biotechnology (2015). DOI: 10.1038/nbt.3210
Benjamin R. Freedman et al. Biomaterials to Mimic and Heal Connective Tissues, Advanced Materials (2019). DOI: 10.1002/adma.201806695
Journal information: Science Advances , The Lancet , Advanced Materials , Nature Biotechnology
© 2020 Science X Network