Common mutation in Parkinson's disease increases cell calcium, may cause brain cell death
Johns Hopkins Medicine researchers have mapped out the cellular pathway that connects the most common genetic mutation associated with Parkinson's disease to brain cell death. In a new study, they show that the mutation initiates a biological pathway that could target brain cells most susceptible to the patterns of cell death leading to Parkinson's disease symptoms.
"This deep dive into the molecular players in Parkinson's disease may provide some answers to its onset and progression," says Valina Dawson, Ph.D., professor of neurology at the Johns Hopkins University School of Medicine, and director of both the neuroregeneration and stem cell programs at the medical school's Institute for Cell Engineering.
The study, published Oct. 1, 2020, in the journal Cell Stem Cell, revealed that a mutation in the leucine-rich repeat kinase 2 (LRRK2) gene shifts the balance of protein production within brain cells, allowing calcium to accumulate inside them. Though mutations in LRRK2 are the most common indicators of inherited Parkinson's disease, their functions within cells are not well understood.
Dawson and her research team learned that a specific mutation within the LRRK2 gene, called Gly2019Ser, or G2019S, produces a protein that becomes more active than normal. They found that this increased activity changes how ribosomes, the protein-making factories of the cell, select which RNA molecules are used to make other proteins. Ribosomes affected by the mutated LRRK2 protein prefer some RNAs with more complex structures than other simple RNAs.
"This preference causes major problems down the line, because it may shift the levels of proteins whose precise regulation is essential for neurons to function and survive," explains Dawson.
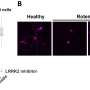
Among the consequences, says Dawson, is the regulation of many cell structures that are responsible for maintaining healthy levels of calcium. One such affected structure is the voltage-gated calcium ion channel, a portal crucial for processing and sending biochemical messages across the brain. Previous studies established that too much calcium in a brain cell can cause it to die. Excess calcium also has been linked as a contributing factor to Alzheimer's disease.
"Mapping out this progression of events is an important advancement in understanding the disease and provides more information on how Parkinson's disease may initially arise," says Dawson.
The research team hopes its findings provide opportunities to create new therapies for Parkinson's disease. "Defining each step in the pathway linking the LRRK2 mutation and brain cell death represents a potential target for drug interaction," says Dawson.
More information: Jungwoo Wren Kim et al. Defects in mRNA Translation in LRRK2-Mutant hiPSC-Derived Dopaminergic Neurons Lead to Dysregulated Calcium Homeostasis, Cell Stem Cell (2020). DOI: 10.1016/j.stem.2020.08.002